Abstract
Download PDF
Full Article
Tea Tree (Melaleuca alternifolia) Fiber as Novel Reinforcement Material for Sugar Palm Biopolymer Based Composite Films
Muhammed L. Sanyang,a Yokasundery Muniandy,b Salit M. Sapuan,a,b,* and Japar Sahari c
The tea tree (Melaleuca alternifolia) is well known for producing essential oil, which is used in medicinal and cosmetic products as a preservative, antiseptic, antibacterial, antifungal, and anti-pest additive. In this study, tea tree residues generated as agro-waste after the tea tree oil extraction process were utilized as cheap fiber material for the reinforcement of sugar palm starch (SPS)-based composite films. The crystallinity and functional groups of tea tree fiber (TTF) were investigated and the effect of TTF loading (0, 1, 3, 5, and 10 wt.%) on the tensile and morphological properties of TTF/SPS composite films were investigated. As the TTF loading increased from 0 to 10 wt.%, the tensile strength and modulus of TTF/SPS composite films were significantly increased, but their elongation at break declined. Optical microscopic and scanning electron microscopic images revealed that the TTF was randomly dispersed in all samples, and there was optimal compatibility between the fiber and matrix. Based on these findings, TTF can be considered as a potential reinforcement material for polymer composite films.
Keywords: Tea tree; Melaleuca alternifolia; Natural fibers; Sugar palm starch; Biocomposites
Contact information: a: Laboratory of Biocomposite Technology, Institute of Tropical Forestry and Forest Products, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia; b: Department of Mechanical and Manufacturing Engineering, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia; c:Faculty of Science and Natural Resources, Universiti Malaysia Sabah, Jalan UMS, 88400 Kota Kinabalu, Sabah, Malaysia; *Corresponding author: sapuan@upm.edu.my
INTRODUCTION
Biocomposites are widely used in the automotive, aerospace, packaging, biomedical, construction, and furniture industries. Biopolymer composites reinforced with natural fiber have gained attention due to their numerous advantages such as affordability, renewability, abundance, environmental friendliness, aesthetics, ease of handling, and an acceptable specific strength and stiffness (Sahari et al. 2012; Sanyang et al. 2016a). The main purpose of developing green biocomposites is to replace the available composites derived from metal, glass, ceramic, wood, and especially non-biodegradable petroleum based plastics (Sain et al. 2005).
Recently, the use of biopolymers in the formulation of packaging films has drastically accelerated. Starch is one of the most widely utilized biopolymers due to its large availability, low cost, and environmental friendliness. Biodegradable films have been prepared using starches from different sources such as cassava, potato, maize, corn, sago, and rice (Sanyang et al. 2015a). Sugar palm starch is another potential biopolymer for the development of biodegradable packaging films (Sahari et al. 2013a,b; Sanyang et al. 2015a,b; 2016b). This starch is obtained from sugar palm trunk especially when the tree is unproductive (Adawiyah et al. 2013). Sugar palm starch has been traditionally used as a raw material for glue substances (Sahari et al. 2012). However, it has not yet been developed into an industrial starch biopolymer.
Despite the many advantages associated with starch-based films, they mostly demonstrate weak mechanical properties, which limit their effective application as a packaging material. To address these hurdles, reinforcement materials such as natural fibers or fillers have been incorporated in the starch matrix to enhance their functional properties. Sahari et al. (2013a) reinforced plasticized sugar palm starch with different sugar palm fiber loading. The mechanical and thermal properties of the sugar palm fiber biocomposites reinforced by plasticized sugar palm starch significantly improved with an increase in the loading of the fibers. Recently, Sanyang et al. (2016b) also reported the effect of sugar palm cellulose in the enhancement of the tensile properties of sugar palm starch-based biocomposite films. In this work, a novel natural fiber reinforcement material (Melaleuca alternifolia (tea tree) fiber) was introduced into the sugar palm starch matrix.
- alternifolia has numerous oil constituents available in the leaf (Brophy and Doran 1996). Although M. alternifolia is native to Australia, mainly found in Queensland stretching to north-east New South Wales, it also extends to neighboring South East Asia and the Pacific (Southwell and Lowe 1999). The Australian aborigines used ground tea tree leaves for wound treatment due to the inherent antiseptic properties of the leaves. They also use tea tree oil as a traditional medicine by inhaling the oils from crushed leaves to treat colds and coughs; they treat skin infections by sprinkling the leaves after applying a poultice (Shemesh and Mayo 1991). Additionally, tea tree leaves can be soaked to make an infusion to treat sore throat and skin ailments. The volatile oil constituents of M. alternifolia led to its commercial development as a medicinal and aromatic plant. This aromatic and medicinal plant genus is popularly recognized for the production of medicinal essential oils. In the form of a formulated or pure oil, tea tree is widely utilized as a preservative, antiseptic, antibacterial, antifungal, or anti-pest additive in an extensive range of medical and cosmetic products (Rodney et al. 2015a). These products include shampoos, conditioners, soaps, bath oils, mouthwashes, toothpastes, deodorants, moisturizers, face washes, foot sprays and powders, shaving products, antiseptic creams, body lotions, sun blocks, lip balms, post-waxing treatments, and acne creams as well as dog shampoos and veterinary care products (Wrigley and Fagg 1993; Southwell and Lowe 1999; Rodney et al. 2015a).
Steam distillation is the most common method for tea tree oil extraction because it causes minimum changes to the composition of the extract and because steam is readily available, cheap, not chemically hazardous, can be used at low pressure, and can be recycled (Davis 1999; Southwell and Lowe 1999). Steam distillation enables the aromatic oil to be extracted at a constant, low temperature that does not damage the oil. In the late 1990s, tea tree essential oil experienced a multi-fold increase in production. During this period, plantation production has risen from an insignificant contribution with respect to bush production to being the predominant source of oil in Australia (Davis 1999; Rodney et al. 2015a). With this development comes the issue of agro-waste from distilled tea tree leaves/branches residue generated after the oil extraction process. High value can be added to such agro-waste by utilizing it as a reinforcement or filler in polymer composites. In this way, the potential environmental impact is averted, and the waste can serve as a cheap reinforcement material to improve the properties of polymer composites.
Rodney et al. 2015b investigated the physical, chemical, and mechanical properties of fibers obtained from different parts of the tea tree such as the trunk, branches, and leaves. From their findings, tea tree trunk fibers manifested the highest cellulose content, which in turn provided the highest tensile strength value compared to their counterparts. Subsequently, Rodney et al. (2015c) utilized tea tree fibers as a reinforcement or filler in tapioca starch composites. The incorporation of 5% tea tree fiber in the tapioca starch matrix enhance the tensile strength of the biocomposite by 203.18% compared to the neat tapioca starch film. Hence, tea tree fibers can be considered as potential reinforcement or filler for thermoplastic starch-based composites. Nevertheless, there has been a limited amount of work done using tea tree fibers as a potential reinforcement material in polymer composites. Therefore, the aim of this work was to determine the effect of tea tree fiber loading on the mechanical properties of sugar palm starch-based films.
EXPERIMENTAL
Preparation of Materials
Tea tree fibers were taken from the Sabah Economic Development & Investment Authority (SEDIA) tea tree field at the Demonstration Plot located at Mile 30 Kimanis, Papar, Sabah, Malaysia. A chainsaw was used to cut down the tree for easy ground harvesting of the fibers.
The fibers then were harvested manually using a knife. Fibers from different parts of the tree such as leaves and twigs were extracted. The tea tree fibers were then dried. The fibers remained unchanged during this process. Sugar palm starch and glycerol were supplied by the Institute of Tropical Forestry and Forest Product (INTROP), University Putra Malaysia.
Characterization of Tea Tree Fibers (TTF)
Infrared (IR) analysis
The main functional groups of the fiber were determined by carrying out Fourier transform infrared (FTIR) spectra analysis. The FTIR analysis was conducted at a resolution of 2 cm-1 from 400 to 4000 cm-1. The sample was mixed with potassium bromide (KBr) to obtain the fiber spectrum.
X-ray diffraction (XRD)
The crystalline structures of TTF were characterized by X-ray diffractometer (APD2000, Italy) with Cu Kα radiation (λ=1.54 Å). The test was conducted under a voltage of 40 kV and a current of 40 Ma. X-ray diffractograms of TTF were recorded for 2θ and the scanning region was between 5° to 40° at scan rate of 2° min-1. The calculation for crystalline index of cellulose (CrI) for treated and untreated fiber was determined based on Segal empirical method stated as follows (Eq. 1),
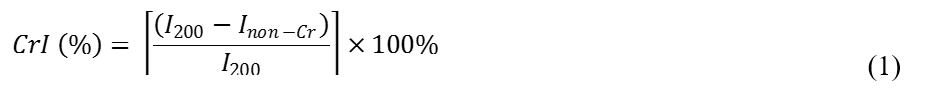
where I200 is the peak intensity corresponding to cellulose and Inon-Cr represents the non-crystalline region.
Fabrication of the Biocomposites
Tea tree fiber-reinforced SPS biocomposites were prepared by solution-casting method (Sanyang et al. 2015a; 2015b; 2016b). Glycerol (G) was used as a plasticizer to reduce the brittleness of SPS. Initially, 8% (w/w) aqueous dispersion of gelatinized sugar palm starch was prepared by heating the film forming solution at 95 °C ± 2 °C. Prior to their casting in glass Petri-dishes, the film forming solutions were cooled. The glass Petri-dishes serving as a casting surface enabled the film to have a smooth and flat surface. The freshly cast films were oven-dried at 40 °C to allow evaporation. After 24 h of drying, films were peeled from the casting surface.
The method was repeated for the fabrication of sugar palm starch biocomposite reinforced with tea tree fibers with different fiber contents (0, 1, 3, 5, and 10 wt.%) (Sanyang et al. 2016b).
Mechanical Properties
A tensile test was carried out in accordance with ASTM D882 (2002) with a universal testing machine (Instron 3365, High Wycombe, England). The crosshead speed was 2 mm/min. A total of 10 samples of 10 mm (W) x 70 mm (L) were tested for different fiber loading biocomposite groups.
Optical Microscopy
An Olympus SZX12 optical microscope (Tokyo, Japan) with a magnification of 7x was used to observe the transparency, porosity, and distribution of tea tree fibers in the TTF/SPS composites. For an alternative view of the surface of tea tree fiber, a high-resolution optical camera was also used for observation.
Scanning Electron Microscopy (SEM)
Scanning electron microscopy (Hitachi S-3400N, Tokyo, Japan) with an operating voltage of 5 kV was used to obtain scanning electron micrographs from the fractured tensile test samples of the biocomposite. The homogeneous distribution of the fibers and the matrix and the adhesion between them was evaluated.
RESULTS AND DISCUSSION
Infrared (IR) analysis of TTF
Figure 1 presents the FTIR spectra of TTF. The broad absorption band at 3286.35 cm-1 is ascribed to the presence of O-H groups, which exist in cellulose, hemicellulose, and lignin of the fiber. The peaks around 2921.14 cm-1 can be attributed to C-H stretching vibration. Similar peaks were reported by Rodney et al. (2015c). The peak found at 1727.01 cm-1 is indicative of the existence of carbonyl groups (C=O) in the hemicellulose and lignin, whereas the intensity peak around 1627 cm-1 is assigned to OH bending of absorbed water within the fiber. In plane CH bending that may be from hemicellulose or cellulose is characterized by the peaks around 1369 cm-1. Similar finding was reported by Fan et al. (2012) while investigating the FTIR spectra of natural fibers.
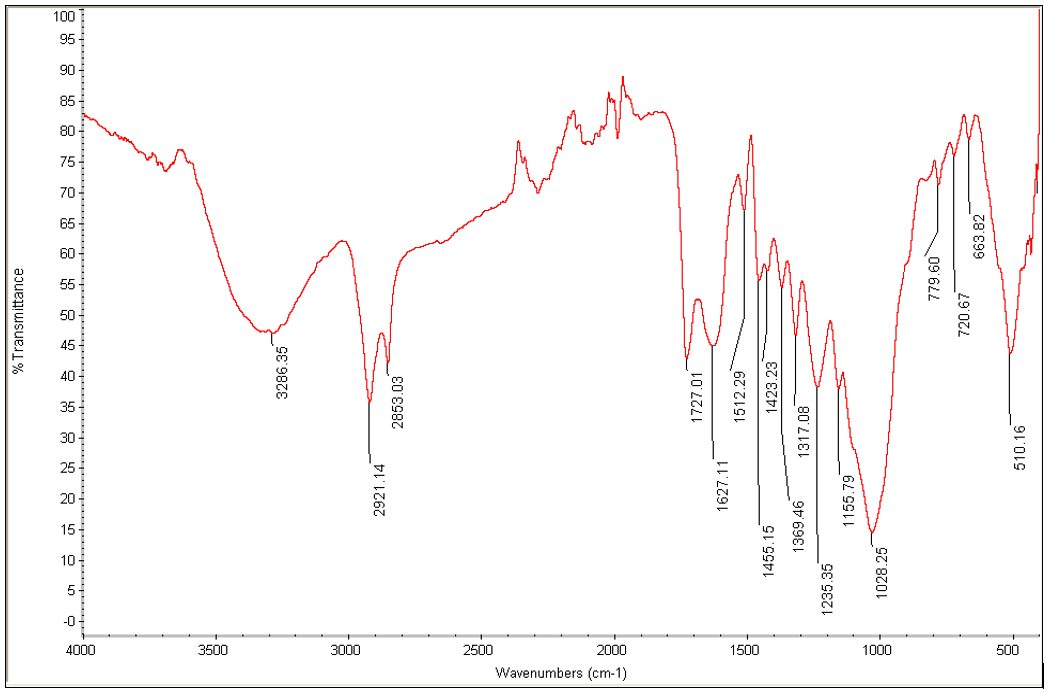
Fig. 1. FTIR spectra of TTF
XRD of TTF
XRD analysis was conducted to determine the crystallinity of TTF. Therefore, high modulus fibers provide better mechanical strength when utilized as reinforcement for polymer composites. The XRD diffractogram of TTF is shown in Fig. 2, where the intensity of the diffraction peak can be clearly observed. The main diffraction peak at 2θ (22.16º) represents a crystalline material which is believed to be the crystallographic planes of cellulose I (I200). Another intensity peak appears near 2θ of 15.12° corresponding to the crystalline phase of cellulose in TTF. On the other hand, the valley shaped region between the two major peaks signifies the non-crystalline region of TTF, which represents the amorphous materials in cellulose, hemicellulose, lignin and other non-cellulose materials. Similar peaks were reported for raw sisal fibers and oil palm empty fruit bunch by Kaushik et al. (2012) and Rayung et al. (2014), respectively. The crystallinity index of TTF is calculated to be 43%. This value is higher than the crystallinity index values reported by Edhirej et al. (2016) and Kaushik et al. (2012) for cassava peel and sisal fiber, respectively. The obtained crystallinity index of TTF is also higher than that of jute (34.3%), kenaf (34.9), and ramie fiber (34.8) reported by Poletto et al. (2014).
Mechanical Properties
Tensile strength
Figure 3 demonstrates the effect of fiber loading on the tensile properties of tea tree fiber-reinforced sugar palm starch (TTF/SPS) biocomposite. The specimen with 10% TTF/SPS content had the highest tensile strength value of 1.223 MPa, followed by 5% TTF/SPS with 1.019 MPa, 3% TTF/SPS with 0.955 MPa, 1% TTF/SPS with 0.761 MPa, and 0% TTF/SPS with 0.646 MPa. The increased tea tree fiber loading improved the tensile strength compared with the neat plasticized SPS thin film by 17.80%, 47.83%, 57.74%, and 89.32% for 1, 3, 5, and 10 wt.% TTF loading, respectively.
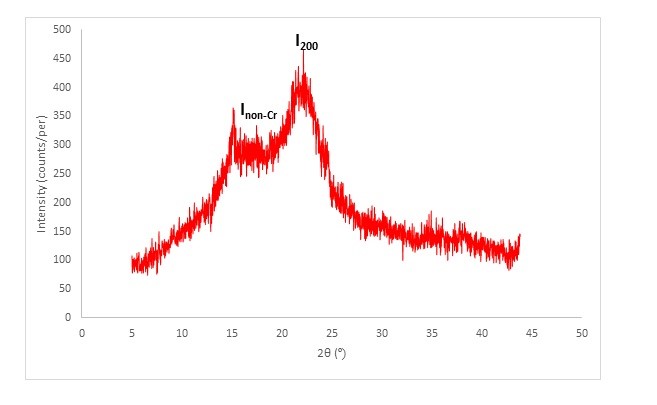
Fig. 2. XRD of TTF
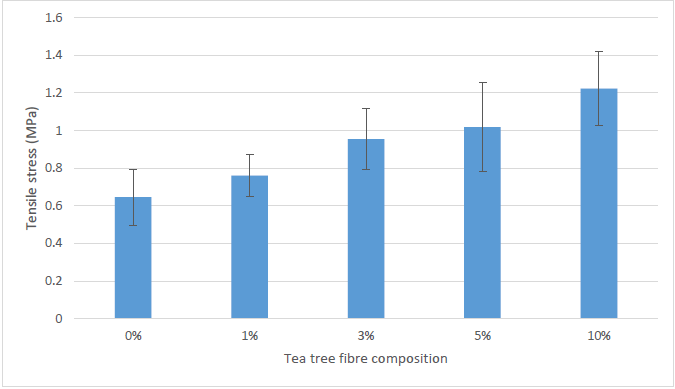
Fig. 3. Tensile strength of the tea tree fiber-reinforced SPS biocomposites
The low fiber loading in 1% TTF/SPS resulted in low tensile strength. This result was attributed to there being insufficient fiber embedded in the SPS matrix to effectively bear and transfer load to neighboring fibers. Thus, the tensile strength of TTF/SPS biocomposites increased with increased tea tree fiber (TTF) loading. The addition of TTF in SPS improved the mechanical strength of the biocomposite material. This result can be attributed to the chemical similarities between the TTF and SPS, which provided good compatibility and strong interfacial adhesion.
These results agree with the findings of many previous studies related to the use of natural reinforcement materials for starch based composite films (Habibi et al. 2008; Sahari et al. 2013a; Sanyang et al. 2016), which supports the observation that the mechanical properties are strongly influenced by the fiber content in the matrix. However, the tensile strength of samples with 5% TTF/SPS were significantly higher as compared to 5% TTF reinforced tapioca starch composites reported by Rodney et al. (2015c). Interestingly, 1% TTF/SPS composite films manifested higher tensile strength value (0.95 MPa) than that of 5% TTF reinforced tapioca starch composites (0.5 MPa). The wide variation in the tensile strength of this comparison can be attributed to the superiority of SPS in film preparation over tapioca starch due to the high amylose content in the native starch of the former as reported by Sanyang et al. (2016a).
Elongation at break (E%)
Figure 4 depicts the E% of TTF/SPS biocomposite films. The E% of TTF/SPS biocomposites decreased as the loading of TTF increased from 0% to 10 %.
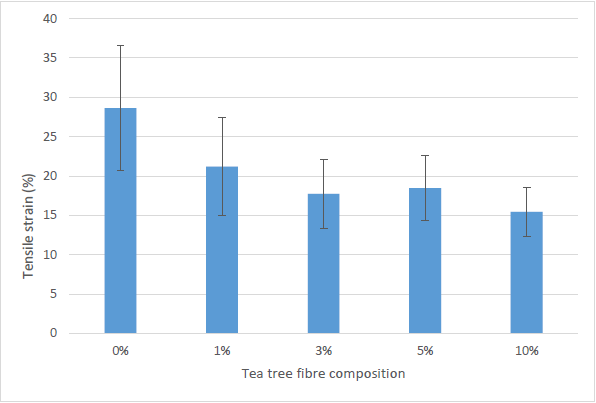
Fig. 4. Tensile at break (strain) of the biocomposite
The addition of the TTF made the E% of TTF/SPS biocomposites decrease from 28.6% to 15.4%. The highest value of elongation at break was observed for 0% TTF/SPS, followed by 1%, 3%, 5%, and 10% TTF/SPS. After the addition of tea tree fiber increases in the SPS matrix, the flexibility of the TTF/SPS composite films decreased by 25.89%, 38.05%, 35.53%, and 46.06% in 1, 3, 5, and 10 wt.% TTF/SPS, respectively.
These results showed that the 0% TTF/SPS biocomposite films were more flexible than the other TTF/SPS biocomposite films. This effect was expected because the incorporation of TTF imparts rigidity and restrains the mobility of the SPS molecules, which lead to an inevitable decrease in the degree of the flexibility of the material. Therefore, the films became less stretchable as their molecular mobility is interrupted by the presence of the TTF. This result concurs with the findings of Sanyang et al. (2016b) for sugar palm starch reinforced with sugar palm cellulose and Edhirej et al. (2016) for bagasse-reinforced cassava starch composite.
Young’s modulus
Figure 5 shows that the 10% TTF/SPS composite had the highest tensile modulus value of 30.50 MPa and also retained the highest stiffness properties. This was followed by 5% TTF/SPS with 20.33 MPa, 3% TTF/SPS with 18.52 MPa, 1% TTF/SPS with 9.86 Mpa, and 0% TTF/SPS with 6.60 MPa.
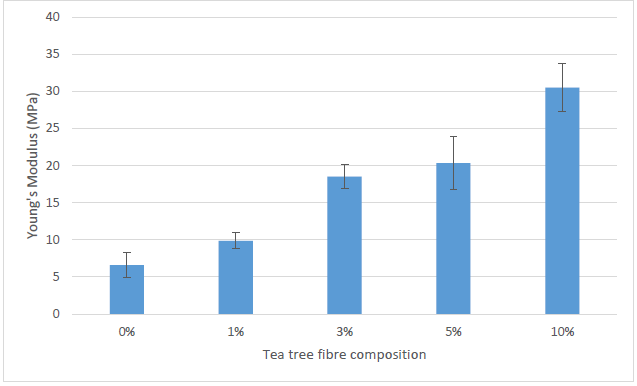
Fig. 5. Young’s Modulus of the tea tree fiber-reinforced sugar palm starch biocomposite
The addition of 1, 3, 5, and 10 wt.% TTF improved the tensile modulus of the neat SPS films by 49.39%, 180.62%, 209.10%, and 362.24%. These results explicitly show that increasing the TTF loading from 1% to 10% led to an increase in the tensile modulus of TTF/SPS composite films. This observed trend can be associated with the weight percentage (wt%) increase of a high stiffness filler (TTF) loaded in a constant weight low stiffness matrix (SPS). Hence, the stiffness of the TTF/SPS biocomposite films significant increased. This result is similar to the previous studies carried out on the loading effect of reinforcement material on the tensile modulus of SPS biocomposites (Sahari et al. 2013a; Sanyang et al. 2016b).
The increase in tensile modulus of TTF/SPS biocomposite can also be attributed to the stronger bonding between the SPS matrix and TTF fiber, which enhances the interfacial adhesion between them, leading to a greater transfer of stress from the matrix to the fibers during tensile testing. The tensile modulus of 5%TTF/SPS (20.33 MPa) composite film is 5 times higher than that of 5 %TTF reinforced tapioca starch (4 MPa) reported by Rodney et al. (2015c). Overall, the mechanical property results observed in the study agreed with those reported by other researchers for natural fibers incorporated in thermoplastic starches (Gáspár et al. 2005; Müller et al. 2009; Sanyang et al. 2016b; Edhirej et al. 2016).
Optical Microscopy
Figure 6 shows optical microscopic images that illustrate the dispersion of TTF in the SPS matrix. More TTF were visible as the TTF loading increased from 1 to 10%. The TTF were randomly dispersed in all samples with the exception of the 0% TTF control film (Fig. 6a). The dispersion of TTF in 1%, 3%, and 5% TTF/SPS composites were random with no significant agglomeration of TTF. However, the addition of 10% TTF loading in the SPS matrix significantly increased the concentration of TTF in the composite film, causing a slight degree of agglomeration, which should have affected their tensile strength. On the contrary, the tensile strength of 10% TTF/SPS composite films increased despite the slight aggregation of TTF. This can be attributed to the good immersion of TTF in the SPS matrix as shown in Fig. 6e, which reflected optimal compatibility between the fiber and matrix.
Scanning electron microscopy
Figure 7 (A-B) shows the plasticized SPS of the fractured portion after the tensile test was performed. Figure 7 (C-D) shows the 1% TTF/SPS biocomposites. The interface between fiber and matrix had poor bonding, as indicated by the wide gap between them. The smooth fracture surfaces had a homogeneous matrix and good adhesion, which improves the mechanical performance of biocomposites (Sahari et al. 2013b).
In both 0% and 1% TTF/SPS SEM micrographs, the smooth surface increase in plasticized SPS was caused by the glycerol. At very low loading of fibers (0 and 1 w/w%), the specimens were very soft, homogeneous, and had smooth surfaces. At higher fiber loading (3, 5, and 10 w/w %), the specimens were fragile, had rough surfaces, and were very brittle.
Figure 7 (E-F) shows the fracture surface of biocomposites with 3% TTF (w/w%). The adhesion in the interface of the fiber and matrix was indicated by the thin gap between them, although voids still existed. The voids in the matrix loosened the fiber-starch bond, and thus the stress was not transferred effectively to the whole composite. This explains why 3% TTF/SPS exhibited a lower tensile strength as compared with 5% and 10% TTF/SPS but was still higher than that of 0% plasticized SPS and 1% TTF/SPS.
The 5% TTF/SPS adhered well to the matrix even though small voids existed (Fig. 7 (G-H)). This was due to the effect of TTF infiltration in the SPS matrix. Figure 7 also depicts fiber breakage, which indicates good adhesion of fiber-matrix interface when subjected to tensile loading. Figure 7 (I-J) shows the 10% TTF/SPS biocomposite, where the TTF adhered to the SPS matrix very well, and TTF was tightly bound to the matrix. TTF embedded into the SPS matrix and the fiber surface was wetted by the SPS matrix, which indicated the phase compatibility. This explains why 10% TTF/SPS exhibited the highest tensile strength, as the bond between the fibers and the starch was strong.

Fig. 6. Image of surface morphology from optical microscopy 7x magnification of tea tree fiber reinforced with plasticized sugar palm starch (SPS) with (A) 0% TTF, (B) 1% TTF, (C) 3% TTF, (D) 5% TTF, and (E) 10% TTF

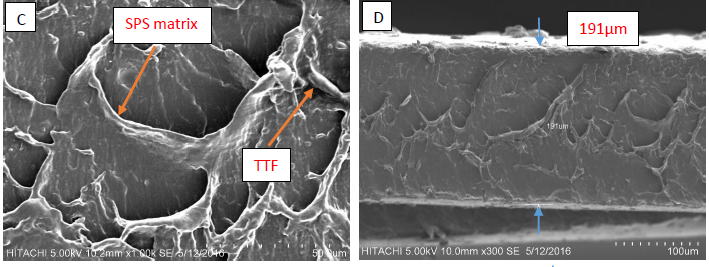



Fig. 7. Scanning electron micrographs of (A) 0%, (C) 1%, (E) 3%, (G) 5%, and (I) 10% with a plasticized SPS cross section of the fracture part after tensile; (B) 0%, (D) 1%, (F) 3%, (H) 5%, and (J) 10% are measures of thin film thickness
CONCLUSIONS
- The FTIR and XRD results manifested that TTF is a potential reinforcement material for SPS due to its OH groups, which can easily form hydrogen bonds with the SPS molecules, whereas its moderate crystallinity is anticipated to generate good fiber tensile strength, which in turn should enhance the mechanical properties of the resulting composites.
- TTF/SPS composite films at different fiber loading were successfully fabricated and characterized.
- All composites showed superior mechanical properties in comparison to neat SPS films. Tensile strength and Young’s modulus were improved, while the tensile strain decreased as the TTF (wt. %) increased.
- SEM and optical microscopy studies showed a good dispersion of the tea tree fiber in the matrix.
- TTF can be effectively used as a reinforcement material in the development of anti-pest biocomposites. It provides a cheap option for the development of green and renewable biocomposites.
ACKNOWLEDGMENTS
The authors thank the Malaysia Ministry of Higher Education for financial support through Research Acculturation Collaborative Effort (RACE) grant.
REFERENCES CITED
ASTM D882 (2002). “Standard test method for tensile properties of thin plastic sheeting,” American Society for Testing and Materials, USA.
Adawiyah, D. R., Sasaki, T., and Kohyama, K. (2013). “Characterization of arenga starch in comparison with sago starch,” Carbohydrate Polymers 92(2), 2306-2313.
Brophy, J. J., and Doran, J. C. (1996). Essential Oils of Tropical Asteromyrtus, Callistemon and Melaleuca Species, Australian Centre for International Agricultural Research (ACIAR), Canberra.
Davis, G. R. (1999). “Tea tree oil distillation,” in: R. Southwell, and I. Lowe (eds.), Tea Tree: The Genus Melaleuca Harwood Academic Publishers, Amsterdam, Netherlands, p. 155.
Edhirej, A., Sapuan, S. M., Jawaid, M., and Ismarrubie Zahari, N. (2016). “Preparation and characterization of cassava starch/peel composite film,” Polymer Composites. DOI: 10.1002/pc.24121
Fan, M., Dai, D., and Huang, B. (2012). “Fourier Transform infrared spectroscopy for natural fibres,” in: Fourier Transform–Materials Analysis, S. M. Salih (ed.), InTech.
Gáspár, M., Benko, Z., Dogossy, G., Réczey, K., and Czigány, T. (2005). “Reducing water absorption in compostable starch-based plastics,” Polymer Degradation and Stability 90(3), 563-569. DOI: 10.1016/j.polymdegradstab.2005.03.012
Habibi, Y., El-Zawawy, W. K., Ibrahim, M. M., and Dufresne, A. (2008). “Processing and characterization of reinforced polyethylene composites made with lignocellulosic fibers from Egyptian agro-industrial residues,” Composites Science and Technology 68(7-8), 1877-1885. DOI: 10.1016/j.compscitech.2008.01.008
Kaushik, V. K., Kumar, A., and Kalia, S. (2012). “Effect of mercerization and benzoyl peroxide treatment on morphology, thermal stability and crystallinity of sisal fibers,” International Journal of Textile Science 1(6), 101-105.
Müller, C. M. O., Laurindo, J. B., and Yamashita, F. (2009). “Effect of cellulose fibers on the crystallinity and mechanical properties of starch-based films at different relative humidity values,” Carbohydrate Polymers 77(2), 293-299. DOI: 10.1016/j.carbpol.2008.12.030
Poletto, M., Ornaghi, H. L., and Zattera, A. J. (2014). “Native cellulose: Structure, characterization and thermal properties,” Materials 7(9), 6105-6119.
Rayung, M., Ibrahim, N. A., Zainuddin, N., Saad, W. Z., Razak, N. I. A., and Chieng, B. W. (2014). “The effect of fiber bleaching treatment on the properties of poly (lactic acid)/oil palm empty fruit bunch fiber composites,” International Journal of Molecular Sciences 15(8), 14728-14742.
Rodney, J., Sahari, J., Mohd Shah, M. K., and Sapuan, S. M. (2015a). “Tea tree (Melaleuca alternifolia) as a new material for biocomposites,” Journal of Applied Science and Agriculture Review 10(3), 21-39.
Rodney, J., Sahari, J., and Mohd Shah, M. K. (2015b). “Physicochemical and mechanical properties of different morphological parts of the tea tree (Melaleuca alternifolia) fibres,” Fibres & Textiles in Eastern Europe 23(6(114)), 31-36. DOI: 10.5604/12303666.1167414
Rodney, J., Sahari, J., Kamal, M., Mohd Shah, M. K., and Sapuan, S. M. (2015c). “Thermochemical and mechanical properties of tea tree (Melaleuca alternifolia) fibre reinforced tapioca starch composites,” e-Polymers 15(6), 401-409.
Sahari, J., Sapuan, S. M., Zainudin, E. S., and Maleque, M. A. (2012). “Sugar palm tree: A versatile plant and novel source for biofibres, biomatrices, and biocomposites,” Polymers from Renewable Resources 3(2), 61-77.
Sahari, J., Sapuan, S. M., Zainudin, E. S., and Maleque, M. A. (2013a). “Mechanical and thermal properties of environmentally friendly composites derived from sugar palm tree,” Materials & Design 49, 285-289.
Sahari, J., Sapuan, S. M., Zainudin, E. S., and Maleque, M. A. (2013b). “Thermo-mechanical behaviors of thermoplastic starch derived from sugar palm tree (Arenga pinnata),” Carbohydrate Polymers 92(2), 1711-1716. DOI: 10.1016/j.carbpol.2012.11.031
Sain, M., Sushara, S., Law, S. S., and Bouilloux, A. (2005). “Interface modification and mechanical properties of natural fiber-polyolefin composite products,” Journal of Reinforced Plastics and Composites 24(2), 121-130. DOI: 10.1177/0731684405041717
Sanyang, M. L., Sapuan, S. M., Jawaid, M. R., and Ishak, J. S. (2015a). “Effect of plasticizer type and concentration on physical properties of biodegradable films based on sugar palm (Arenga pinnata) starch for food packaging,” Journal of Food Science and Technology, 1-11.
Sanyang, M. L., Sapuan, S. M., Jawaid, M., Ishak, M. R., and Sahari, J. (2015b). “Effect of plasticizer type and concentration on dynamic mechanical properties of sugar palm starch based films,” International Journal of Polymer Analysis and Characterization 20(7), 627-636. DOI: 10.1080/1023666X.2015.1054107
Sanyang, M., Sapuan, S., Jawaid, M., Ishak, M., and Sahari, J. (2015c). “Effect of plasticizer type and concentration on tensile, thermal and barrier properties of biodegradable films based on sugar palm (Arenga pinnata) starch,” Polymers 7(6), 1106-1124. DOI: 10.3390/polym7061106
Sanyang, M. L., Sapuan, S. M., Jawaid, M., Ishak, M. R., and Sahari, J. (2016a). “Recent developments in sugar palm (Arenga pinnata) based biocomposites and their potential industrial applications: A review,” Renewable and Sustainable Energy Reviews 54, 533-549. DOI: 10.1016/j.rser.2015.10.037
Sanyang, M. L., Sapuan, S. M., Jawaid, M., Ishak, M. R., and Sahari, J. (2016b). “Effect of sugar palm-derived cellulose reinforcement on the mechanical and water barrier properties of sugar palm starch biocomposite films,” BioResources 11(2), 4134-4145. DOI: 10.15376/biores.11.2.4134-4145
Shemesh, A., and Mayo, W. L. (1991). “Australian tea tree oil: A natural antiseptic and fungicidalagent,” The Australian Journal of Pharmacy. Journal of Pharmaceutical Sciences 72, 802-803.
Southwell, I., and Lowe (1999). Tea Tree: The Genus Melaleuca. Amsterdam, Netherlands: Harwood Academic Publishers.
Wrigley, J. W., and Fagg, M. (1993). Bottlebrushes, Paperbarks & Tea Trees, Angus & Robertson, Sydney.
Article submitted: December 6, 2016; Peer review completed: February 14, 2017; Revised version received: March 22, 2017: Accepted: March 25, 2017; Published: April 4, 2017.
DOI: 10.15376/biores.12.2.3751-3765
