Abstract
The present study describes the development of a new bioadsorbent from lignocellulosic wastes of agricultural origin. The biosorption capacity of an agricultural solid waste, pine bark (Pinus densiflora Sieb.), to remove phenolic compounds (phenol, 2-chlorophenol (2-CPh), and 4- chlorophenol (4-CPh)) from aqueous solutions under batch equilibrium conditions was investigated. The morphological characteristics of the biosorbent were evaluated by BET surface area analysis, Fourier transform infrared spectroscopy (FTIR), elemental analysis, an X-ray diffractometer (XRD), and a scanning electron microscope (SEM). Batch experiments were conducted to investigate the effect of initial pH (2 to 10), contact time, initial concentration of adsorbate (50 to 200 mg/L), and biosorbent dosage. The biosorption of phenolic compounds decreased with increasing pH, and the highest biosorption capacity was achieved at a pH of 6.0. Biosorption equilibrium was established in 120 min. The biosorption equilibrium data were fitted and analyzed with Langmuir, Freundlich, and Dubinin-Radushkevich isotherm equations, as well as four adsorption kinetic models. The kinetics data fitted well into the pseudo-second-order kinetic model, with a correlation coefficient greater than 0.993. The maximum monolayer biosorption capacity of pine bark for phenol, 2-CPh, and 4-CPh was found to be 142.85, 204.08, and 263.15 mg/g, respectively, as calculated by the Langmuir model at 30 ± 1 °C. Pine bark could be used as a new effective, low-cost biosorbent material with good uptake capacity and rapid kinetics for the removal of phenolic compounds from aqueous media.
Download PDF
Full Article
Biosorption of Phenolic Compounds from Aqueous Solutions using Pine (Pinus densiflora Sieb) Bark Powder
Nadavala Siva Kumar,a,* Hasfalina Che Man,a, and Heung-Sik Woo b
The present study describes the development of a new bioadsorbent from lignocellulosic wastes of agricultural origin. The biosorption capacity of an agricultural solid waste, pine bark (Pinus densifloraSieb.), to remove phenolic compounds (phenol, 2-chlorophenol (2-CPh), and 4- chlorophenol (4-CPh)) from aqueous solutions under batch equilibrium conditions was investigated. The morphological characteristics of the biosorbent were evaluated by BET surface area analysis, Fourier transform infrared spectroscopy (FTIR), elemental analysis, an X-ray diffractometer (XRD), and a scanning electron microscope (SEM). Batch experiments were conducted to investigate the effect of initial pH (2 to 10), contact time, initial concentration of adsorbate (50 to 200 mg/L), and biosorbent dosage. The biosorption of phenolic compounds decreased with increasing pH, and the highest biosorption capacity was achieved at a pH of 6.0. Biosorption equilibrium was established in 120 min. The biosorption equilibrium data were fitted and analyzed with Langmuir, Freundlich, and Dubinin-Radushkevich isotherm equations, as well as four adsorption kinetic models. The kinetics data fitted well into the pseudo-second-order kinetic model, with a correlation coefficient greater than 0.993. The maximum monolayer biosorption capacity of pine bark for phenol, 2-CPh, and 4-CPh was found to be 142.85, 204.08, and 263.15 mg/g, respectively, as calculated by the Langmuir model at 30 ± 1 °C. Pine bark could be used as a new effective, low-cost biosorbent material with good uptake capacity and rapid kinetics for the removal of phenolic compounds from aqueous media.
Keywords: Biosorption; Pine bark powder; Phenolic compounds; Kinetics and isotherm models
Contact information: a: Department of Biological and Agricultural Engineering, Faculty of Engineering, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor Darul Ehsan, Malaysia; b: Department of Safety Environmental System Engineering, Dongguk University, Gyeongju 780-714, Republic of Korea;
*Corresponding authors: shivanadavala@gmail.com; hasfalina@upm.edu.my
INTRODUCTION
Phenols and chlorophenols are contaminants in soils, sediments, surface waters, and groundwater, because of their worldwide utilization in the last 50 years. In recent years, there have been increasing environmental and toxicological interests about phenolic pollutants due to their widespread occurrence and relative frequency in the aquatic environment (McKinney et al. 2006; Zeng et al. 2008). The main sources of phenol and chlorophenols are refineries, petrochemical industries (Kumar et al. 2011; Sanjay et al. 2008), industrial resins, plastics, adhesives, rubber, iron, steel, aluminum, pulp and paper industries, pesticides, fungicides, bactericides, herbicides, medical and health products (including oils, softeners, mouthwash, and eye and ear drops), tannins, electrical industries, and paint industries (Kumar et al. 2009a; Srivastava et al. 2006; Verschueren 2009). Phenol and its derivatives are considered noxious pollutants because they are toxic and harmful to living organisms, even at low concentrations. Phenol is a colorless solid and easily miscible in water, so it cannot be identified in water through the naked eye. The United States Environmental Protection Agency (USEPA) has listed phenolic compounds as priority compounds. Stringent US Environmental Protection Agency (EPA) regulation calls for lowering phenol content in the wastewater to less than 1 mg/L (Banat et al. 2000).The utilization of phenol-contaminated waters causes protein degeneration, vomiting, tissue erosion, smoky colored urine, paralysis of the central nervous system, and damage to the kidney, liver, and pancreas in human bodies (Knop and Pilato 1985). Because of the low biological degradability, high toxicity, and high ecological persistence of phenolic compounds, wastewater containing phenolic compounds must be treated before being discharged to receiving media (Bayramoglu et al. 2009).
To keep waters free from phenol compounds, various processes have been employed for the removal of phenols from aqueous media, including chemical oxidation (He et al. 2009), membrane filtration (Kujawski et al. 2004), biodegradation (El-Naas et al. 2009), electro coagulation (Ugurlu et al. 2008), photo degradation (Gomez et al. 2011), solvent extraction (Xu et al. 2006), and adsorption (Yang et al. 2008). Among these methods, adsorption is still the most popular and widely used technique for phenol removal because of its simple design, easy operation, and relatively simple regeneration (Nevskaia et al. 2004). This has led many researchers to search for more cost-effective and efficient adsorbents to remove organic contaminants from water and wastewater. Fly ash (Aziz et al. 2014; Batabyal et al. 1995), rice husk (Mona et al. 1997), peat (Viraraghavan and Alfaro 1998), bentonite (Senturk et al. 2009), and polymeric adsorbents (Li et al. 2002; Pan et al. 2005) have been tested for the adsorption of organic pollutants. Currently, very innovative and cost effective methods are used for the removal of toxic substances from waste waters.
Biosorption, as an efficient, cost-effective, and environmentally friendly technique for heavy metals and various organic pollutants, has emerged as a potential alternative to conventional techniques (Febrianto et al. 2009). The search for new and innovative treatment techniques has focused attention on the adsorption capacities of other adsorbents, such as agricultural by-products and lignocellulosic residues (Garg et al. 2009; Rao and Viraraghavan 2002), which are readily available and do not need to be regenerated due to their low cost. Recent studies on the sorption of metals and organic pollutants can be removed using plant materials such as Acacia leucocephala bark (Kumar and Min 2011b; Kumar et al. 2012), Pinus pinaster bark (Vazquez et al. 2002), pine bark (Bras et al. 1999), formaldehyde pretreated Pinus pinaster bark (Vazquez et al. 2007), and lignocellulosic substrate extracted from wheat bran (Dupont et al. 2003).
Pine bark, an agricultural and easily available solid waste, could be an alternative for more costly wastewater treatment processes. Pine (Pinus densiflora) belongs to the family Pinaceae and is widely distributed around the world, including East-Asian countries such as Korea, Japan, and China. Pine is a popular lumber for furniture, paper, plywood, and crafts. It is a soft wood and can dent or scratch easily, making it more popular for house framing and plywood than for fine furniture. Most utility poles are also pine trees. It can be burned for fire wood, especially when a quick fire is needed, as in cooking or a fire pit. The majority of the waste bark is used as a low-grade thermal fuel. The bark is a waste product that has a variety of uses. All mature pine trees put on an annual layer of bark, which contributes to the increasing diameter of the stems. In some species, the outermost layer dies and is annually deciduous either in long strips or in variably sized flakes. Because of the large number of pine trees in Korea, a massive amount of bark is produced, which is disposed of as waste. Lignocellulosic wastes such as tree bark, lignin, wood, conifer leaves, sawdust, peanut skin, and hull have been considered low-cost and popular biosorbents to remove metals from wastewater, as these wastes are readily available in bulk at almost no cost (Aoyama et al. 2000; Sciban et al. 2007; Shukla et al. 2005). Pine bark can be an attractive and important biomass resource, accounting for about 10 to 15% of the total tree weight (Kofujita et al. 1999). The kinetics of the adsorption of metal and uranyl ions onto amidoximated Pinus densiflora bark was studied by Mun et al. (2010). To the best of our knowledge, there are no reports on the application of this biosorption study for the removal of phenolic compounds from aqueous solutions. The aim of the present work is to study the removal of phenolic compounds by pine bark material from synthetic wastewater and to offer this biosorbent as a local substitute for existing commercial adsorbent material.
Therefore, the main objective of this present research work is to investigate the biosorption potentiality of pine bark biosorbent material for the removal of phenol, 2-chlorophenol (2-CPh), and 4-chlorophenol (4-CPh) from aqueous medium. Further, the biosorbent was characterized by Brunauer-Emmett-Teller (BET) surface area, Fourier transform infrared spectroscopy (FTIR), scanning electron microscopy (SEM), X-ray diffractometer (XRD), and elemental analysis for carbon, hydrogen, and nitrogen (CHN) to identify the functional groups, crystallinity and surface morphology, and elemental mapping. The effects of factors such as contact time, biosorbent dosage, pH, and initial adsorbate concentrations on this biosorption process were investigated using a batch equilibrium technique.
MATERIALS AND METHODS
Materials
Pine bark (Pinus densiflora Sieb.) was collected from the campus woods at Dongguk University, Gyeongju, South Korea, in June, 2011, for the preparation of biosorbent. The required raw materials, phenol (Junsei Chemical Co. Ltd.; Tokyo, Japan), 2-CPh, and 4-CPh (Junsei Chemicals Co.; Korea), were used without further purification. Stock solutions were prepared by dissolving 1.0 g of phenol, 2-CPh, and 4-CPh individually in one liter of double distilled water. These stock solutions were used to prepare 50, 100, 150, and 200 mg/L solutions of phenol, 2-CPh, and 4-CPh. To adjust pH, 0.1 M HCl and 0.1 M NaOH were obtained from Samchun Chemicals, South Korea. Water used for the preparation of solutions and cleaning adsorbents was generated in the laboratory by double distilling deionized water in a quartz distillation unit.
Characterization
X-ray diffraction (XRD) measurements were carried out using a D5000 (Siemens; Germany) diffractometer equipped with a copper anode (Cu Kα = 1.5406 Å) over a scanning interval (2θ) value ranging from 10 to 50°. Elemental (CHN) analyses were recorded using a Thermo Finnigan Flash EA 1112 CHN analyzer (Thermo Fisher Scientific, USA) for the pine bark powder biosorbent. The BET surface area of the pine bark powder was determined on a QUADRASORB analyzer (Quantachrome Instruments; USA) by nitrogen adsorption at -196 °C. Fourier transform infrared spectra of pine bark powder, in virgin form and loaded with phenolic compounds, were obtained using a Nicolet-740, Perkin-Elmer model 283B (USA). The sample was prepared as a KBr pellet by investigating the peaks within the range 4000−400 cm−1. A variable pressure SEM (Model Evo MA 15; Carl Zeiss; England) was used to characterize the surface morphology of the pine bark powder. The sample was analyzed using the back-scattered electrons, and images were then obtained at 800x and 1000x magnification with an accelerating voltage of 10.0 kV.
Preparation of Biosorbent
The local pine tree bark was first washed with distilled water to remove mud and dirt and then dried for 48 h in an oven at 60 °C. Then, the bark material was ground into a very fine particle size. The fine pine bark was soaked in 0.1 M NaOH to remove lignin materials, followed by soaking in 0.1 M H2SO4. Finally, it was washed with distilled water several times and dried in an oven at 70 to 80 °C for 6 to 9 h and cooled at room temperature in desiccators. The dried pine bark powder was stored in desiccators until used. The dried bark was sieved to 55- to 75-μm mesh with a standard testing sieve and used as a biosorbent for phenolic compound adsorption.
Batch Adsorption Studies
To optimize the adsorption of phenolic compounds, the effect of experimental conditions such as pH, contact time, biosorbent dosage, and initial adsorbate concentration were studied. In adsorption equilibrium, experiments were conducted in a set of 125-mL Erlenmeyer flasks, where 100-mL solutions of phenolic compounds (phenol, 2-CPh, and 4-CPh) with different initial concentrations (50 to 200 mg/L) were added to these flasks. The initial pH of the solution was adjusted using 0.1 M HCl/NaOH solution. Pine bark powder (0.1 g) was added to the phenolic compound solutions, and each sample was kept in a shaking water bath at 30 ± 1 °C and 220 rpm for 120 min to reach equilibrium. Samples were taken from the tubes, and the solutions were separated from the adsorbent by filtering through Whatman No. 50 filter paper (2.7 µm particle retention) to eliminate any fine particles. Then, the concentration of phenolic compounds was determined by measuring absorbance using a 1601 UV/Vis spectrophotometer (Shimadzu; Japan) at 270 nm, 274 nm, and 280 nm for phenol, 2-CPh, and 4-CPh, respectively. The amount of adsorption at equilibrium, qe (mg/g) was calculated from the following equation:
(1)
where C0 and Ceq (mg/L) are the initial and equilibrium liquid-phase concentrations of the phenolic compounds, respectively. V (L) is the volume of the solution, and m is the mass (g) of the dry pine bark powder used. For batch kinetic studies, the same procedure was followed, but the aqueous samples were taken at preset time intervals. The concentrations of phenolic compounds were similarly measured. The amount of uptake at time t, qt (mg g-1), was calculated as:
(2)
where Ct (mg/L) is the liquid-phase concentration of phenolic compounds at time t (min).
RESULTS AND DISCUSSION
Characterization of the Biosorbent
Fourier transform infra-red (FTIR), X-ray diffraction (XRD), scanning electron microcopy (SEM), elemental (CHN), and surface area by nitrogen adsorption (BET) analyses were carried out to characterize the pine bark powder. Physicochemical characterization of pine bark was performed to understand the mechanism involved in the biosorption process. The moisture content, bulk density, ash content, CHN, and surface properties (such as surface area and pore radius) are presented in Table 1.
Table 1. Physical, Surface Characterization, and Elemental Analysis of Pine Bark Powder Biosorbent
FTIR Analysis
Characterization of the biosorbent was carried out by Fourier Transform Infrared Spectroscopy to determine the type of functional groups on the biosorbent. FTIR spectroscopy was used to obtain information on the nature of possible adsorbent-adsorbate interactions. FTIR spectra in the range of 4000 to 400 cm−1 for the virgin pine bark powder and pine bark powder loaded with phenol, 2-CPh, and 4-CPh are shown in Fig. 1, profiles (a), (b), (c), and (d), respectively. The FTIR spectroscopic characteristics are shown in Table 2. The FTIR spectrum of pine bark powder before adsorption (Fig. 1a) shows a broad absorption peak at 3372 cm-1 corresponding to the overlapping -OH and -NH peaks. A peak at 2921 cm-1 represents the C-H group. The C=O stretching of pure pine bark powder was at 1617 cm−1. The presence of –C–N linkages is confirmed from the peaks at 1058 cm−1. As shown in Fig. 1b, c, and d, a significant difference can be seen in the FTIR spectra of biosorbent before and after biosorption. Some peaks were slightly shifted and/or broadened, indicating that the functional groups present on the biosorbent are involved in interaction with the phenolic compounds. These results confirm the participation of the amino, carboxyl, and hydroxyl groups of pine bark powder as potential active binding sites for the adsorption of phenol, 2-CPh, and 4-CPh.
Fig. 1. FTIR spectra of pine bark powder: (a) before biosorption; (b) after phenol biosorption; (c) after 2-CPh biosorption; and (d) after 4-CPh biosorption
Table 2. Wave Number (cm−1) for the Dominant Peak from FTIR for Phenolic Compounds Adsorption
XRD Analysis
An X-ray diffraction analysis was performed on virgin pine bark powder. The pine bark powder exhibited a broad peak at 2θ = 29.8° (figure not shown), indicating the amorphous state of the biosorbent.
Scanning Electron Microscope (SEM) Studies
The morphology of the pine bark powder consisted of a brick type, plate-like structure (Fig. 2a) with an agglomerated and irregular surface structure. The coarse surfaces of pine bark powder have an irregular structure, indicating that it has high porosity, thus making it possible for the adsorption of phenol, 2-CPh, and 4-CPh on different parts of the biosorbent (Fig. 2b).
Fig. 2. Scanning electron micrographs of the pine bark powder at (a) 1000x and (b) 800x magnification
Effect of pH
The pH is the most important parameter affecting the degree of ionization and the allocation of various pollutants, leading to changes in the reaction kinetics and equilibrium characteristics of the sorption process. To optimize the pH to achieve maximum removal efficiency, experiments were conducted over a wide range of pH, from 2.0 to 10.0, using 0.1 g of pine bark with 100 mL of 100 mg/L adsorbate solutions at 30 ± 1 °C. In the alkaline range, the pH was varied using aqueous 0.1 M NaOH, whereas in the acidic range, the pH was varied using 0.1 M HCl. Experimental results indicate that phenol, 2-CPh, and 4-CPh were most effectively removed by pine bark at pH 6.0; the removal efficiency decreased on either side of these pH values (Fig. 3).
Fig. 3. Effect of pH on the biosorption of () phenol, () 2–CPh, and () 4-CPh onto pine bark powder. Experimental conditions: initial concentrations = 100 mg/L, contact time = 3 h, biosorbent dosage = 0.1 g/0.1 L, temp = 30 ± 1 °C, and agitation rate = 220 rpm.
The effect of solution pH on the removal efficiency of phenol and its derivatives from aqueous solutions can be explained by considering the presence of ionic and molecular forms of phenolic compounds in aqueous solutions. The phenolic compounds considered in this study, i.e., phenol, 2-CPh, and 4-CPh, have pKa values of 9.9, 8.3, and 9.2, respectively, suggesting that phenol is a weak acid; hence, they mostly exist as anions at high pH values (Kumar and Min 2011a). When the pH of a solution surpasses the pKa, phenols chiefly exist as negative phenolate ions, whereas they exist as neutral molecules below the pKa. Because of the electron-rich nature of the oxygen atom in phenolate ions, the hydrogen bonding efficiency decreases. Therefore, phenols are effectively adsorbed on to the adsorbent as neutral molecules, not phenolate ions. From the experimental results, pH 6.0 was selected as the optimum pH value.
Effect of Biosorbent Dosage
The effect of different doses of pine bark on the removal of phenol, 2-CPh, and 4-CPh was determined, and the results are presented in Figs. 4a through 4c. The amount of adsorbent was varied from 0.05 to 0.8 g while variables such as pH, rpm, contact time, and temperature were kept constant.
Fig. 4. Effect of biosorbent dosage on the biosorption of (a) phenol, (b) 2-CPh, and (c) 4-CPh onto pine bark powder [(% removal of phenol, 2-CPh, 4-CPh, and biosorption capacity (mg/g)]. Experimental conditions: initial concentration = 100 mg/L, biosorbent dosage = 0.05 to 0.8 g, contact time = 120 min, and pH = 6.0.
It can be seen from the figures that the percentage removal of phenol, 2-CPh, and 4-CPh increased with increasing adsorbent dose, while the loading capacity, qe (mg/g), (amount of phenol, 2-CPh, and 4-CPh loaded per unit weight of adsorbent) gradually decreased. This growth can be attributed to the additional number of adsorption sites, which resulted from the increase in the adsorbent dosage. The decrease in the total adsorbed amount of phenols (qe) as the adsorbent dose increased can be attributed to the aggregation or overlap of the adsorption sites due to the overcrowding of adsorbent particles, which decreases the total adsorbent surface area available to the phenols. It can also be seen from Figs. 4a through 4c that the solute uptake markedly increased to an adsorbent dose of 0.5 g/0.1 L; thereafter, no significant increase was observed. The binding rate of phenolic compounds with adsorbent increases more rapidly in the initial stages; after a certain point, the adsorption is marginal and becomes almost constant.
Effect of Contact Time and Initial Concentration
Contact time and initial concentration have a pronounced effect on the removal of adsorbate species from aqueous solutions. The effect of agitation time on the extent of adsorption of phenolic compounds at different concentrations is shown in Figs. 5, 6, and 7 for phenol, 2-CPh, and 4-CPh, respectively.
Fig. 5. Effect of contact time on phenol adsorption. Different initial phenol concentrations () C0 = 50 mg/L, (●) C0 = 100 mg/L, (▲) C0 = 150 mg/L, () C0 = 200 mg/L; pH = 6.0; biosorbent dosage = 0.1 g/0.1 L; contact time = 120 min; agitation rate = 220 rpm; temp = 30 ± 1 °C
The effect of shaking time on the biosorption of phenolic compounds onto pine bark powder was studied over a range of 20 to 180 min, using 0.1 g/0.1 L of pine bark powder, 50 to 200 mg/L of phenolic compounds at pH 6.0, 30 ± 1 °C, and a shaking speed of 220 rpm. The saturation curves rise sharply in the initial stages, indicating that there are plenty of readily accessible sites. Eventually, a plateau is reached in all curves, indicating that the adsorbent is saturated at this level.
It can be seen from figures that the contact time needed for phenol solutions with initial concentrations of 50 to 200 mg/L to reach equilibrium was 120 min. Almost no remarkable improvement was observed after longer contact time. After this equilibrium period, the amount of solute adsorbed did not change significantly with time, indicating that this time is sufficient to attain equilibrium for the maximum removal of phenolic compounds from aqueous solutions by pine bark powder. Therefore, the optimum contact time was selected as 120 min for further experiments.
However, for adsorbate solutions with higher initial concentrations, lower equilibrium times were required. An increase in initial adsorbate solution concentrations resulted in increased phenolic compounds uptake. The removal curves are single, smooth, and continuous, indicating the formation of monolayer coverage of the phenol molecules onto the outer surface of the adsorbent.
Fig. 6. Effect of contact time on 2-CPh adsorption. Different initial 2-CPh concentrations () C0 = 50 mg/L, (●) C0 = 100 mg/L, (▲) C0 = 150 mg/L, () C0 = 200 mg/L; pH = 6.0; biosorbent dosage = 0.1 g/0.1 L; contact time = 120 min; agitation rate = 220 rpm; temp = 30 ± 1 °C
Fig. 7. Effect of contact time on 4-CPh adsorption. Different initial 4-CPh concentrations () C0 = 50 mg/L, (●) C0 = 100 mg/L, (▲) C0 = 150 mg/L, () C0 = 200 mg/L; pH = 6.0; biosorbent dosage = 0.1g/0.1 L; contact time = 120 min; agitation rate = 220 rpm; temp = 30 ± 1 °C
KINETIC STUDIES
Several kinetic models (the model of diffusion on homogeneous surface, the model of the diffusional pores, the heterogeneous model of diffusion, and the model of Elovich) were tested on our experimental results to describe the mechanism of the process of adsorption (mass transfer, chemical reaction). Four kinetic models, pseudo-first order, pseudo-second order, Elovich, and the intraparticle diffusion model, were considered to investigate the behavior of the adsorption process of phenolic compounds onto pine bark.
The pseudo-first-order kinetic model was suggested by Lagergren and is widely used for the description the sorption of liquid on solid (Lagergren 1898). The linear form of the pseudo-first-order rate equation is generally expressed as follows,
(4)
where qe(mg/g) and qt(mg/g) are the amounts of adsorbate sorbed at equilibrium and at time t (min). k1is the rate constant of the first-order adsorption process. The pseudo-first order kinetic constants were determined from slope of the plot of log (qe− qt) versus t (figure not shown), and the values are shown in Table 2.
The kinetic data were further analyzed using Ho’s pseudo-second-order kinetic model (Ho et al. 1999). The linearized form of the equation is expressed as:
(5)
The values of qe and k2 can be estimated experimentally from the slope and intercept of the plot of t/qtversus t (figure not shown) and are given in Table 2. Adsorption of phenol, 2-CPh, and 4-CPh on pine bark satisfactorily followed the pseudo-second order rate equation. This was reflected by the high R2values (> 0.993) and the agreement between both the calculated and the experimental qe values (Table 3).
To quantitatively compare the applicability of each model, a normalized standard deviation![]() was calculated, defined as,
was calculated, defined as,
(6)
where the subscripts ‘exp’ and ‘cal’ refer to the experimental and calculated values, respectively, and N is the number of data points. The lower the value of ![]() , the better fit the model is for the data. The calculated rate constants for the models, their corresponding regression (R2), and normalized standard deviation values are listed in Table 3. The results suggest that the higher R2 values obtained with the pseudo-second-order kinetic model, lower
, the better fit the model is for the data. The calculated rate constants for the models, their corresponding regression (R2), and normalized standard deviation values are listed in Table 3. The results suggest that the higher R2 values obtained with the pseudo-second-order kinetic model, lower![]() values, and the calculated qe (cal) values are closer to the experimental data than the calculated values of pseudo-first order kinetic model. From this observation it may be concluded that the adsorption of phenol, 2-CPh, and 4-CPh compounds on the pine bark powder follows pseudo-second order kinetics.
values, and the calculated qe (cal) values are closer to the experimental data than the calculated values of pseudo-first order kinetic model. From this observation it may be concluded that the adsorption of phenol, 2-CPh, and 4-CPh compounds on the pine bark powder follows pseudo-second order kinetics.
Table 3. Biosorption Rate Constants of Phenol, 2-CPh, and 4-CPh on Pine Bark Powder
Elovich Equation
The Elovich equation was first used in the kinetics of adsorption of gases on solids; it has been successfully applied for the adsorption of solutes from a liquid solution. The linear form of the Elovich equation is given as follows (Bulut et al. 2008):
(7)
where a (mg/g min) is the initial sorption rate and the parameter b (g/mg) is a constant related to the extent of surface coverage and activation energy for chemisorptions. The parameters (1/b) and (1/b)ln(ab) can be obtained from the slope and intercept of the linear plot of qt versus lnt. The plots are linear, with good correlation coefficient values (Table 4). The calculated qe values from the Elovich model agreed quite well with the experimental equilibrium concentration values. This suggests that the sorption system studied belongs to the pseudo-second order kinetic model, based on the assumption that the rate-determining step may be chemisorption, involving valence forces through sharing or exchange of electrons between the adsorbent and adsorbate.
Intraparticle Diffusion Study
In diffusion studies, the rate can be expressed in terms of the square root of time. The mathematical dependence of Qt versus t1/2 is obtained if the process is considered to be influenced by diffusion in the particles and convective diffusion in the solution. According to the intraparticle diffusion model proposed by Weber and Morris (1963), the root time dependence may be expressed by the following equation:
(8)
where Qt is the amount of solute on the surface of the sorbent at time t (mg/g), kid is the intraparticle diffusion rate constant (mg/g min1/2), and C is the intercept (mg/g); this gives an idea of the thickness of the boundary layer.
The kid values are found from the slopes of Qt versus t1/2 plots and are shown in Figs. 8a through 8c. If the intraparticle diffusion is involved in the adsorption process, then the plot of the square root of time versus the uptake (Qt) would result in a linear relationship, and the intraparticle diffusion would be the controlling step if this line passed through the origin.
When the plots do not pass through the origin, this is indicative of some degree of boundary layer control, which further shows that the intraparticle diffusion is not the only rate controlling step; other processes may control the rate of adsorption. From Table 4, it can be seen that the thickness of the boundary layer increased with increasing concentration.
Fig. 8. Weber–Morris model plots for biosorption of (a) phenol, (b) 2-CPh, and (c) 4-CPh onto pine bark powder
Table 4. Biosorption Rate Constants of Phenol, 2-CPh, and 4-CPh on Pine Bark Powder
EQUILIBRIUM ADSORPTION ISOTHERM MODELS
The capacity of the adsorption isotherm is fundamental and plays an important role in the determination of the maximum adsorption capacity. Three isotherm equations have been tested in the present study to analyze the equilibrium data of the adsorbent: Langmuir, Freundlich, and Dubinin-Radushkevich. Analysis of the adsorption equilibrium data was done on the widely used Langmuir and Freundlich adsorption isotherms equations, which provide information on the relationship between the quantity of adsorbate removed and the concentration of the adsorbate remaining in the liquid phase at equilibrium.
The Langmuir theory was first used to describe the adsorption of gas molecules on solid surfaces. However, it has since been successfully applied to mass transfer processes from liquid to solid phase for describing the mechanism of adsorption. Theoretically, it depicts a monolayer uptake of the adsorbate on a homogenous adsorbent surface having uniform binding energy at all binding sites. The Langmuir (Kumar and Min 2011a) adsorption equation is expressed as:
(9)
where Ce (mg/L) is the equilibrium concentration of the adsorbate, qe (mg/g) is the amount of adsorbate adsorbed per unit mass of adsorbent, and Q0 and b are Langmuir constants related to the adsorption capacity and rate of adsorption, respectively. The linear plot of 1/Ce versus 1/qe indicates that adsorption obeys the Langmuir model.
The Langmuir parameters can be further used to predict the affinity between the adsorbate and adsorbent using the dimensionless separation factor (RL) (Kumar and Min 2011b) as follows,
(10)
where RL is the dimensionless separation factor, C0 is the initial concentration of the adsorbate (mg/L), and b is the Langmuir constant (L/mg). The RL parameter is considered a more reliable indicator of the adsorption. The RL value predicts the fate of the adsorption system, as favorable (0 > RL > 1), unfavorable (RL > 1), linear (RL = 1), or irreversible (RL = 0). The value of RL is less than 1 and greater than 0, suggesting the favorable uptake of phenol, 2-CPh, and 4-CPh compounds by pine bark powder.
The Freundlich isotherm model (Kumar et al. 2012) is an empirical description based on the adsorption on a heterogeneous surface containing binding sites having different energies, represented as,
(11)
where KF ((mg/g)(L/mg)1/n) and (1/n) are Freundlich constants that indicate relative adsorption capacity and adsorption intensity, respectively, and n > 1 indicates favorable adsorption. The linear form of Eq. 9 is,
(12)
which will have a straight line with a slope of 1/n and an intercept of ln(KF) when ln(qe) is plotted against ln(Ce).
The Dubinin-Radushkevich (1947) isotherm was used to fit with experimental data, and it can be represented as,
(13)
where qs is the Dubinin-Radushkevich monolayer capacity (mg/g), B is a constant related to sorption energy, and ε is the Polanyi potential, which is related to the equilibrium concentration as follows:
(14)
where R is the gas constant (8.314 J/mol K) and T is the absolute temperature. The constant B gives the mean free energy E of adsorption per molecule of the adsorbate when it is transferred to the surface of the solid from infinity in the solution and can be computed using the relationship:
(15)
A plot of ln(qe) versus ɛ2 enables the constants qs and E to be determined. E gives information about the nature of adsorption mechanism, whether it is a chemical ion exchange or a physical adsorption. If 8 < E < 16 kJ/mol, then the adsorption process is a chemical ion-exchange process. If E < 8 kJ/mol, then the adsorption process is physical in nature. Correlation coefficients and parameter values for the three isotherms were presented in Table 5. Based on the correlation coefficients, the applicability of the isotherms was compared. For all cases, the Langmuir equation fits the experimental data better than the Freundlich and Dubinin-Radushkevich equation. All the evaluated equilibrium models had a good fit with the experimental data, and it can be concluded that the Langmuir is the best model describing adsorption of phenolic compounds on the pine bark powder, as it gave the maximum R2 value among the considered models. The comparison of maximum monolayer adsorption capacities of different adsorbents obtained from different sources are listed in Table 6 along with the values obtained in the present study.
Table 5. Isotherm Parameters of Langmuir, Freundlich, and D-R Isotherms for Adsorption of Phenol, 2-CPh, and 4-CPh on Pine Bark Powder
Table 6. Maximum Adsorption Capacities, Q0 (mg/g), for the Adsorption of Phenol, 2-CPh and 4-CPh Compounds by Various Adsorbents
CONCLUSIONS
- Lignocellulosic agriculture waste, i.e., pine bark powder, was utilized as a biosorbent for the removal of phenolic compounds (phenol, 2-CPh, and 4-CPh) from aqueous environments.
- The biosorbent was characterized by FTIR, XRD, BET, CHN analyzer, and SEM techniques.
- The adsorption rate process was fast, reaching equilibrium in a short time (almost 120 min).
- When the pine bark biosorbent dosage was increased, the equilibrium adsorption capacity (mg/g) of pine bark gradually decreased, whereas the percent removal efficiency increased.
- The good correlation coefficients and the agreement between experimental and calculated values of qe and lower
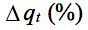 values showed that the phenolic compounds adsorption followed pseudo-second order kinetics.
values showed that the phenolic compounds adsorption followed pseudo-second order kinetics. - The experimentally determined Q0max of pine bark powder for phenol, 2-CPh, and 4-CPh was 142.85, 204.08, and 263.15 mg/g, respectively, which agree well with the predicted Langmuir Q0max values. The experimental equilibrium data could be well interpreted by the Langmuir isotherm. The adsorption of phenolic compounds was rated as favorable, based on RL values.
- Considering the present findings, it can be concluded that pine bark powder, an abundantly and locally available low-cost biosorbent, has a considerable adsorption capacity with the potential for use as an alternative sorbent for the removal of phenol, 2-CPh, and 4-CPh compounds from aqueous media.
REFERENCES CITED
Aoyama, M., Tsuda, M., Cho, N.-S., and Doi, S. (2000). “Adsorption of trivalent chromium from dilute solution by conifer leaves,” Wood Science and Technology 34(1), 55-63.
Aziz, A. S. A., Latifah, A. M., Hasfalina, C. M., and Kumar, N. S. (2014). “Kinetic modelling and isotherm studies for copper (II) adsorption onto palm oil boiler mill fly ash (POFA) as natural low-cost adsorbent,” BioResources 9(1), 336-356.
Banat, F. A., Al-Bashir, B., Al-Asheh, S., and Hayajneh, O. (2000). “Adsorption of phenol by bentonite,” Environmental Pollution 107(3), 391-398.
Batabyal, D., Sahu, A., and Chaudhuri, S. K. (1995). “Kinetics and mechanism of removal of 2,4-dimethyl phenol from aqueous solutions with coal fly-ash,” Separations Technology 5(4), 179-186.
Bayramoglu, G., Gursel, I., Tunali, Y., and Arica, M. Y. (2009). “Biosorption of phenol and 2-chlorophenol by Funalia trogii pellets,” Bioresource Technology 100(10), 2685-2691.
Bras, I. P., Santos, L., and Alves, A. (1999). “Organochlorine pesticides removal by Pinus bark sorption,” Environmental Science and Technology 33(4), 631-634.
Bulut, E., Ozacar, M., and Sengil, I. A. (2008). “Equilibrium and kinetic data and process design for adsorption of Congo Red onto bentonite,” Journal of Hazardous Materials 154(1-3), 613-622.
Denizli, A., Cihangir, N., Rad, A. Y., Taner, M., and Alsancak, G. (2004). “Removal of chlorophenols from synthetic solutions using Phanerochaete chrysosporium,” Process Biochemistry 39(12), 2025-2030.
Denizli, A., Cihangir, N., Tuzmen, N., and Alsancak, G. (2005). “Removal of chlorophenols from aquatic systems using the dried and dead fungus Pleurotus sajor caju,” Bioresource Technology 96(1), 59-62.
Dubinin, M. M., and Radushkevich, L. V. (1947). “Equation of the characteristic curve of activated charcoal,” Chemisches Zentralblatt 1(1), 875-889.
Dupont, L., Bouanda, J., Dumonceau, J., and Aplincourt, M. (2003). “Metal ions binding onto a lignocellulosic substrate extracted from wheat bran: a NICA–Donnan approach,” Journal of Colloid and Interface Science 263(1), 35-41
El-Naas, M. H., Al-Muhtaseb, S. A., and Makhlouf, S. (2009). “Biodegradation of phenol by Pseudomonas putida immobilized in polyvinyl alcohol (PVA) gel,” Journal of Hazardous Materials,164(2-3), 720-725.
Febrianto, J., Kosasih, A., Sunarso, J., Ju, Y., Indraswati, N., and Ismadji, S. (2009). “Equilibrium and kinetic studies in adsorption of heavy metals using biosorbent: A summary of recent studies,” Journal of Hazardous Materials 162(2-3), 616-645.
Garg, V. K., Gupta, R., Bala, A., and Kumar, R. (2009). “Dye removal from aqueous solution by adsorption on treated sawdust,” Bioresource Technology 89(2), 121-124.
Gomez, M., Murcia, M. D., Gomez, E., Gomez, J. L., and Christofi, N. (2011). “Degradation of phenolic pollutants using KrCl and XeBr excilamps in the presence of dye: A comparative study,” Desalination 274(1-3), 156-163.
He, D., Guan, X., Ma, J., and Yu, M. (2009). “Influence of different nominal molecular weight fractions of humic acids on phenol oxidation by permanganate,” Environmental Science and Technology 43(21), 8332-8337.
Ho, Y. S., and McKay, G. (1999). “Pseudo-second order model for sorption processes,” Process Biochem 34(5), 451-465.
Knop, A., and Pilato, L. A. (1985). Phenolic Resins: Chemistry, Applications and Performance, Springer-Verlag, Berlin.
Kofujita, H., Ettyu, K., and Ota, M. (1999). “Characterization of the major components in bark from five Japanese tree species for chemical utilization,” Wood Science and Technology 33(3), 223-228.
Kujawski, W., Warszawski, A., Ratajczak, W., and Capala, W. (2004). “Removal of phenol from waste water by different separation techniques,” Desalination 163(1-3), 287-296.
Kumar, N. P., Kumar, N. S., and Krishnaiah, A. (2012). “Defluoridation of water using tamarind (Tamarindus indica) fruit cover: Kinetics and equilibrium studies,” Journal of the Chilean Chemical Society 57(3), 1224-1231.
Kumar, N. S., and Min, K. (2011a). “Phenolic compounds biosorption onto Schizophyllum communefungus: FTIR analysis, kinetics, and adsorption isotherms modeling,” Chemical Engineering Journal168(2), 562-571.
Kumar, N. S., and Min, K. (2011b). “Removal of phenolic compounds from aqueous solutions by biosorption onto Acacia leucocephala bark powder: Equilibrium and kinetic studies,” Journal of the Chilean Chemical Society 56(1), 539-545.
Kumar, N. S., Boddu, V. M., and Krishnaiah, A. (2009). “Biosorption of phenolic compounds by Trametes versicolor polyporus fungus,” Adsorption Science & Technology 27(1), 31-46.
Kumar, N. S., Reddy, A. S., Boddu, V. M., and Krishnaiah, A. (2009b). “Development of chitosan alginate based biosorbent for the removal of p-chlorophenol from aqueous medium,” Toxicological and Environmental Chemistry 91(6), 1035-1054.
Kumar, N. S., Subbaiah, M. V., Reddy, A. S., and Krishnaiah, A. (2009a). “Biosorption of phenolic compounds from aqueous solutions onto chitosan-abrus precatorius blended beads,” Journal of Chemical Technology and Biotechnology 84(7), 972-981.
Kumar, N. S., Suguna, M., Subbaiah, M. V., Reddy, A. S., Kumar, N. P., and Krishnaiah, A. (2010). “Adsorption of phenolic compounds from aqueous solutions onto chitosan-coated perlite beads as biosorbent,” Industrial and Engineering Chemistry Research 49(19), 9238-9247.
Kumar, N. S., Woo, H. S., and Min, K. (2012). “Equilibrium and kinetic studies on biosorption of 2,4,6-trichlorophenol from aqueous solutions by Acacia leucocephala bark,” Colloids and Surfaces B-Biointerfaces 94(1), 125-132.
Lagergren, S. (1898). “Zur theorie der sogenannten adsorption gelöster stoffe,” Kungliga Svenska Vetenskapsakademiens. Handlingar 24(4), 1-39.
Li, A. M., Zhang, Q. X., Zhang, G. C., Chen, J. L., Fei, Z. H., and Liu, F. Q. (2002). “Adsorption of phenolic compounds from aqueous solutions by a water-compatible hypercrosslinked polymeric adsorbent,” Chemosphere 47(9), 981-989.
McKinney, M. A., Cesh, L. S., Elliott, J. E., Williams, T. D., Garcelon, D. K., and Letcher, R. J. (2006). “Brominated flame retardants and halogenated phenolic compounds in North American west coast bald eaglet (Haliaeetus leucocephalus) plasma,” Environmental Science and Technology 40(20), 6275-6281.
Mona, E., Zein, R., Kurniadi, R., and Kurniadi, I. (1997). The use of rice husk for removal of phenol from waste water as studied using 4-aminoantipyrine spectrophotometric method,” Environmental Technology 18(3), 355-358.
Mun, S. P., Ku, C. S., and Kim, J. P. (2010). “Adsorption of metal and uranyl ions onto amidoximated Pinus densiflora bark,” Wood Science and Technology 44(2), 283-299.
Nevskaia, D. M., Castillejos-Lopez, E., Munoz, V., and Guerrero-Ruiz, A. (2004). “Adsorption of aromatic compounds from water by treated carbon materials,” Environmental Science and Technology38(21), 5786-5796.
Pan, B. C., Zhang, X., Zhang, W. M., Zheng, J. Z., Pan, B. J., Chen, J. L., and Zhang, Q. X. (2005). “Adsorption of phenolic compounds from aqueous solution onto a macroporous polymer and its aminated derivative: Isotherm analysis,” Journal of Hazardous Materials 121(1-3), 233-241.
Rao, J. R., and Viraraghavan, T. (2002). “Biosorption of phenol from an aqueous solution by Aspergillus niger biomass,” Bioresource Technology 85(2), 165-171.
Rubin, E., Rodriguez, P., Herrero, R., and Sastre de, Vicente, M. (2006). “Biosorption of phenolic compounds by the brown alga Sargassum muticum,” Journal of Chemical Technology and Biotechnology 81(7), 1093-1099.
Sciban, M., Radetic, B., Kevresan, Z., and Klasnja, M. (2007). “Adsorption of heavy metals from electroplating wastewater by wood sawdust,” Bioresource Technology 98(2), 402-409.
Senturk, H. B., Ozdes, D., Gundogdu, A., Duran, C., and Soylak, M. (2009). “Removal of phenol from aqueous solutions by adsorption onto organomodified Tirebolu bentonite: Equilibrium, kinetic, and thermodynamic study,” Journal of Hazardous Materials 172(1), 353-362
Shukla, S. S., Yu, L. J., Dorris, K. L., and Shukla, A. (2005). “Removal of nickel from aqueous solutions by sawdust,” Journal of Hazardous Materials 121(1-3), 243-246.
Siva Kumar, N., Kalyani, S., Boddu, V. M., and Krishnaiah, A. (2008). “Biosorption of phenol and o-chlorophenol from aqueous solutions on to chitosan calcium alginate blended beads,” Journal of Hazardous Materials 162(1), 482-489.
Srivastava, V. C., Swamy, M. M., Mall, I. D., Prasad, B., and Mishra, I. M. (2006). “Adsorptive removal of phenol by bagasse fly ash and activated carbon: Equilibrium, kinetics and thermodynamics,” Colloids Surface A: Physicochemical Engineering Aspects 272(1-2), 89-104.
Ugurlu, M., Gurses, A., Dogar, C., and Yalcin, M. (2008). “The removal of lignin and phenol from paper mill effluents by electro coagulation,” Journal of Environmental Management 87(3), 420-428.
Vazquez, G., Gonzalez-Alvarez, J., Freire, S., Lopez-Lorenzo, M., and Antorrena, G. (2002). “Removal of cadmium and mercury ions from aqueous solution by sorption on treated Pinus pinaster bark: Kinetics and isotherms,” Bioresource Technology 82(3), 247-251.
Vazquez, G., Gonzalez-Alvarez, J., Garcia, A. I., Freire, M. S., and Antorrena, G. (2007). “Adsorption of phenol on formaldehyde-pretreated Pinus pinaster bark: Equilibrium and kinetics,” Bioresource Technology 98(8), 1535-1540.
Verschueren, K. (2009). Handbook of Environmental Data on Organic Chemicals, John Wiley, New York.
Viraraghavan, T., and Alfaro, F. D. (1998). “Adsorption of phenol from wastewater by peat, fly ash and bentonite,” Journal of Hazardous Materials 57(1-3), 59-70.
Weber, W. J., and Morris, J. C. (1963). “Kinetics of adsorption on carbon from solution,” Journal of Sanitary Engineering Division. American Society of Civil Engineering 89(2), 31-60.
Xu, J. Q., Duan, W. H., Zhou, X. Z., and Zhou, J. Z. (2006). “Extraction of phenol in waste-waters with annular centrifugal contactors,” Journal of Hazardous Materials, 131(1-3), 98-102.
Yang, K., Wu, W., Jing, Q., and Zhu, L. (2008). “Aqueous adsorption of aniline, phenol, and their substitutes by multi-walled carbon nanotubes,” Environmental Science and Technology 42(21), 7931-7936.
Zeng, G. M., Xu, K., Huang, J. H., Li, X., Fang, Y.Y., and Qu, Y. H. (2008). “Micellar enhanced ultrafiltration of phenol in synthetic wastewater using polysulfone spiral membrane,” Journal of Membrane Science 310(1-2), 149-160.
Article submitted: February 27, 2014; Peer review completed: June 16, 2014; Revised version received and accepted: July 3, 2014; Published: July 14, 2014.
