Abstract
Stabilized fertilizers that contain nitrification inhibitors and/or urease inhibitors are widely used in China. A pot experiment was conducted to analyze soil enzymatic characteristics related to carbon and nitrogen turnover and metabolism under the use of rice straw and stabilized fertilizer. Results showed that stabilized fertilizer exhibited the highest yield production, panicle numbers, and above-ground biomass. Compared with urea treatment with straw, adding inhibitors reduced soil organic carbon and the enzyme activity related to acquisition of carbon, but increased soil organic carbon accumulation, rice yield, and above-ground biomass. Stabilized fertilizer increased protease activity; however, it decreased N-acetyl-β-glucosaminide. Addition of straw significantly increased dissolved organic and microbial biomass carbon or nitrogen, as well as the enzyme activities of α-D-glucosidase, β-D-glucosidase, β-N-acetyl-glucosidase, and cellulase at the seedling and tillering stages. The principal components analysis showed that the synthesis of extracellular enzyme related to carbon and nitrogen acquiring act as a proxy for straw decomposing under nitrogen conditions. The combination delayed the release of ammonia, which affected the carbon and nitrogen coupling by microbial organisms. These results demonstrated a relationship between soil carbon and nitrogen dynamics and soil enzymes in different fertilization management.
Download PDF
Full Article
Effect of a Urea and Urease/Nitrification Inhibitor Combination on Rice Straw Hydrolysis and Nutrient Turnover on Rice Growth
Chunxiao Yu,a,b Lili Zhang,a,* Lijie Yang,a,c Wei Bai,d Chen Feng,d Wentao Li,a,b Kaikuo Wu,a,b Dongpo Li,a and Zhijie Wu a
Stabilized fertilizers that contain nitrification inhibitors and/or urease inhibitors are widely used in China. A pot experiment was conducted to analyze soil enzymatic characteristics related to carbon and nitrogen turnover and metabolism under the use of rice straw and stabilized fertilizer. Results showed that stabilized fertilizer exhibited the highest yield production, panicle numbers, and above-ground biomass. Compared with urea treatment with straw, adding inhibitors reduced soil organic carbon and the enzyme activity related to acquisition of carbon, but increased soil organic carbon accumulation, rice yield, and above-ground biomass. Stabilized fertilizer increased protease activity; however, it decreased N-acetyl-β-glucosaminide. Addition of straw significantly increased dissolved organic and microbial biomass carbon or nitrogen, as well as the enzyme activities of α-D-glucosidase, β-D-glucosidase, β-N-acetyl-glucosidase, and cellulase at the seedling and tillering stages. The principal components analysis showed that the synthesis of extracellular enzyme related to carbon and nitrogen acquiring act as a proxy for straw decomposing under nitrogen conditions. The combination delayed the release of ammonia, which affected the carbon and nitrogen coupling by microbial organisms. These results demonstrated a relationship between soil carbon and nitrogen dynamics and soil enzymes in different fertilization management.
Keywords: Enzyme mechanisms; C and N turnover; Stabilized fertilizer; Paddy soil; Rice straw
Contact information: a: Institute of Applied Ecology, Chinese Academy of Sciences, No. 72 Wenhua Road, Shenyang 110016, China; b: University of Chinese Academy of Sciences, No. 15A, Yuquan Road, Beijing 100049, China; c: Shenyang Research Institute of Chemical Industry, No. 8 Liaodong Road, Shenyang 110021, China; d: Liaoning Academy of Agricultural Sciences, No. 84 Dongling Road, Shenyang 110161, China; *Corresponding author: llzhang@iae.ac.cn
GRAPHICAL ABSTRACT
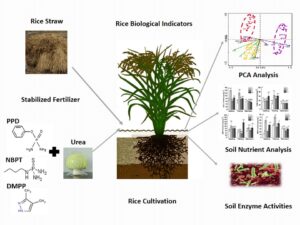
INTRODUCTION
In China, agricultural residues are essentially a renewable biomass resource, and returning crop residues to field is a common agricultural management strategy (Lu et al. 2018). Straw biomass has been used as coating material for the controlled release of fertilizers into the soil, and possesses high density, good degradability, and superior controlled release properties (Lu et al. 2015; Chen et al. 2017). Research has shown that the addition of straw influences the nitrogen (N) uptake efficiency (Cucu et al. 2013). There is a great potential of straw to be combined with stabilized fertilizer; however there has been a need for research in this area. Stabilized fertilizers refers to fertilizers with urease inhibitors and/or nitrification inhibitors added during the production process (Wu et al. 2019).
Applying stabilized fertilizer has been demonstrated to be an effective agricultural management practice for N-fertilizer use, which reduces the loss of gas emission through prohibiting urea release and the nitrification process, increases the content of NH4+-N available for plant uptake, and then influences the nitrogen turnover of soil (Shi et al. 2015; Meng et al. 2020). In addition to the effect on N-transformation, research also shows that N-fertilization affects the rate of soil organic carbon (SOC) decomposition through regulating extracellular enzyme activities (Jian et al. 2016). Wang et al. (2019) considered that fertilization regimes had a significant influence on microbial communities and soil enzyme activities.
Soil enzymes play a key role in nutrient cycling, as well as the utilization of carbon (C) and nitrogen (N). They also contribute to the stock and export of soil dissolvable organic carbon (DOC) and dissolvable organic nitrogen (DON) pools (Zhang et al. 2015), and also correlate with soil microbial biomass (Wang et al. 2019). Cellulases are a group of hydrolytic enzymes that soil microbes produce to decompose polysaccharides. They include α-1,4-glucosidase (AG), β-D-glucosidase (BG), β-D-cellulobiosidase (CEL), and β-1,4-xylosidase (XYL). Glucosidase acts as a release agent for small molecular sugars, providing potential energy for microbes. The activity of α-glucosidase and β-glucosidase play an important role in the decomposition of cellulose of soil. Glucosidase is an important component of cellulolytic enzyme system (Fujita et al. 2018). The cellulase hydrolyzes cellulose into monosaccharides, which indicates the ability of soil to decompose plant residues. Kader et al. (2017) reported that glucosidase and cellulase are involved in the accumulation and conversion of organic C and that the hydrolysis of the terminal NH2 group in amino acid is the limiting step of soil N-mineralization. In addition, the enzymes associated with microbial N-acquisition include β-1,4-N-acetyl-glucosaminidase (NA) and protease (PG) (Jian et al. 2016). Additionally, the N-acetyl-glucosidase degrades chitin into soluble subunits that can be absorbed and utilized by soil microorganisms (Yang et al. 2016).
Soil organic carbon (SOC) is the aggregate of humus, animal and plant residues, and microorganisms formed by microbial action. Soil dissolvable organic C (DOC) refers to the organic carbon that can be lost through a membrane with an aperture of 0.45 microns without evaporation during the analysis. Microbial biomass and soil enzymes work together as catalysts in the conversion of soil organic C to soluble organic C (Eagle et al. 2000; Bowles et al. 2013) and are also used as indicators of changes in soil properties induced by soil management (Kandeler et al. 1999), especially the C and N materials input. Straw added into paddy soil along with exogenous C mainly consists of protein and cellulose; and chitin is present in soils acting as fungal and macro faunal residue, thus increasing the reaction of soil substrate and the enzyme source. The enzyme correlating to chitin and cellulose is an important factor to characterize straw decomposition (Wang et al. 2016). As the available N source, especially the inorganic N source, and the living environment of soil microorganisms improves, it triggers increased soil enzyme activity (Geisseler and Scow 2014). The stabilized fertilizer coupled with straw N also affects the key extracellular enzyme activities and the turnover of C and N in paddy soil, and then affects the rice N adsorption and yield.
The enzyme activity helps to identify soil C and N conversions and kinetics after the addition of rice straw and the stabilized fertilizer in the northeast paddy soil. Therefore, it is essential to explore the relationship between the change of enzyme activity and C or N conversion. To date, most researchers have aimed at straw management and normal N fertilizer (Shi et al. 2015; Sihi et al. 2019), but few have focused on stabilized fertilizer with a new kind of inhibitors combination and rice straw. Hence, this study explored the impact of straw and stabilized fertilizer added on soil C and N turnover according to related enzymes and rice growth. Furthermore, this work considered the potential of rice straw and stabilized urea on straw hydrolysis and urea transformation. This provides theoretical data for straw and inhibitor combination to slow down nitrogen release and improve microbial activity, which accelerates the development of stabilized fertilizer of straw film coating and fertilization strategy in paddy fields.
EXPERIMENTAL
Experimental Site
An outdoor pot experiment was conducted at Shenyang National Agricultural Ecosystem Field Observation and Research Station (41°31 ‘N, 123°24’ E) located in Shenyang, China. The tested paddy soil was taken from 0 to 20 cm soil layer of the experimental site (Shenyang Agricultural University Rice Research Institute). Soil was shielded at 5 mm to remove stones, crop stubble, and roots. The soil contained 20.43 g C per kg and 1.85 g N per kg soil, with a C/N of 11.04 and pH of 6.18. Rice straw comprised of 37.8 g C per kg and 5.96 g N per kg, with C/N ratio of 63.
Experimental Design
Four treatments were set in this experiment, i.e., urea (N), urea plus straw (NS), stabilized urea (NI), and stabilized urea plus straw (NIS). Each treatment had three replicates; there were 36 pots in total and they were randomly arranged, and flood-cultivated for 120 days. Each pot was a plastic basin with a straight configuration. The diameter was 18 cm, and the height was 20 cm. Each pot was filled with 3 kg dry soil. Potassium chloride (KCl) and calcium superphosphate (CaP2H4O8) were applied as basal fertilizer, with 100 mg P per kg soil and 150 mg K per kg soil. Stabilized urea was added in amounts of 60, 45, and 45 mg N per kg soil before culture, seedling stage, and tillering stage, respectively.
The new NI sample included urease plus inhibitor combination, which were 1% phenyl phosphorodiamidate (PPD) (Ourchem, Sinopharm Chemical Reagent Co. Ltd., Shanghai, China), 1% thiophosphoric triamide (NBPT) (Ourchem, Sinopharm Chemical Reagent Co., Ltd., Shanghai, China), and 2% 3,4-dimethylpyrazole phosphate (DMPP) (Maya Reagent Biotech, Jiaxing, China). The straw was added at 5 g/kg, which is equivalent to straw yield in a season. Meifeng 9 (Oryza sativa L.) was selected in this study, which is planted widely in northeast China. Crop management was performed consistently with local conventional farming practices.
The rice plant and soil were sampled after the seedling stage (June 4, 2018), tillering stage (June 25, 2018), and maturity stage (September 21, 2018). Seedling stage was about 7 days after rice plant transplanted and basal fertilizer application, which is the peak period that urea nitrogen hydrolysis. Tillering stage was the most vigorous in nitrogen metabolism in rice life and rice straw hydrolysis. It is important both in terms of nitrogen transformation and soil nutrient accumulation. To detect the effect of different nitrogen and straw on soil C and N accumulation, the soil and rice plant were evaluated after harvest.
Methods
Rice plants were harvested at three sampling times. In order to inhibit the respiration of plants, avoid the loss of biomass, and reduce the measurement error of biomass, the plants were green retarded at 85 ℃ for 2 h, then oven-dried until constant weight at 65 ℃, and ground to powder to detect the total nitrogen content (Elementar, Thermo, Heraeus, Germany). The ammonia (NH4+-N) and nitrate (NO3–-N) contents were analyzed using 2 mol/L KCl (100 mL), which was digested with 20 g fresh soil, and determined with a continuous flow analyzer at the wavelengths of 660 nm and 540 nm (AA3; Bran Luebbe, Heidelberg, Germany) (Yang et al. 2016). Microbial biomass C and N were determined by chloroform fumigation method (Joergensen 1996; Joergensen and Mueller 1996). The ratio of soil to extract liquid used was 1:4. Microbial biomass nitrogen (MBN) was measured using a Vario TOC Select Analyzer (Elementar, Heraeus, Germany). The fumigation coefficient N was 0.54, and C was 0.45. Determination of soil dissolved organic C and N: the extracted solution was filtered through 0.45-um filter membrane and determined by an elemental analyzer (TOC). Protease activity was determined by the Ladd and Butler (1972) method for control and substrate samples, the control was added after the soil sample incubation and prior to analysis. The extracellular enzyme activities of α-D-glycosidase enzymes (AG), β-D-glycosidase enzymes (BG), N-acetyl-β-glucosaminide enzymes (NA), β-D-xyloside (XYL), and β-D-cellobioside enzyme (CEL) were determined as described with colorimetric determination methods described by Marx et al. (2001). Briefly, 1.0 g fresh soil was homogenized with 100 mL 50 mM acetate buffer (pH = 5.0), 10 µM 4-methyl umbrella ketone as a reference and 200 µM substrates were dispensed into the black 96-well microplate, then covered and incubated at 2 ℃ for 4 h in the dark. The reaction was stopped by adding 50 μL 0.5M NaOH. The fluorescence was determined immediately in a fluorometer (BMG Labtech, Offenburg, Germany) at 360 nm excitation and 460 nm emission.
Statistical Analysis
All statistical analysis were conducted by Excel (Microsoft, Office 2010, Washington, USA) and SPSS 16.0 software (SPSS Inc., SPSS16.0, Chicago, USA), one-way analysis of variance (ANOVA) with Duncan’s method was performed for nutrient contents and soil enzyme activities at different management practice (p < 0.05). Principal component analysis (PCA) analysis was conducted by Canoco 5 (Micro-computer Power, Ithaca, New York, USA) with data represented by mean value (n = 3). Origin 8.0 (OriginLab Inc, Origin 8.0, Guangzhou, China) was used for drawing.
RESULTS AND DISCUSSION
Nitrogen Uptake and Utilization by Rice Plants
According to the results of rice biomass and nitrogen adsorption, as shown in Table 2, no significant effect was observed at the seedling stage (P > 0.05), while N and NI samples indicated significantly higher biomass at the tillering stage (P < 0.05). However, at the maturation stage, NS harvest provided the worst yield, biomass, N adsorption, and the number of panicles. The rice biomass and yields of N, NI, and NIS samples increased 23.0%, 27.8%, 22.8%, and 16.4%, 31.2%, 25.2%, respectively, compared with NS. The worst effect of urea with organic material might be because of the amount of carbon source addition that made the competition of plant and microbial more intense (Inselsbacher et al. 2010). It also increased NH3 volatilization and thus reduced the available N for rice growth (San et al. 2011). The NI and NIS had the highest yield, panicle numbers, and above-ground biomass. Silva et al. (2017) showed that NBPT increased crop yield 5.3%; the yield was significantly higher with the inhibitor combinations in this experiment (P < 0.05). In addition, the stabilized urea alleviated the yield reduction compared with urea plus straw. The NIS had the highest N adsorption content; the results were constant with Ma (2015) in the upland soil. When both the agronomic and economic effects were considered, the stabilized urea and coupled with rice straw were recommended.
Effects of Stabilized Fertilizer and Straw on Soil NH4+-N and NO3–-N Contents
Soil inorganic nitrogen is the preferred form that the microorganisms and rice would be absorbing. According to Fig. 1, all treatments showed a negative trend of NH4+-N content at seedling stage. The application of straw immobilized more NH4+ ions and NI showed an inhibitory function of nitrification, which slowed down the process of urea hydrolysis and nitrification, reduced the release of NH4+ions. Although the trend was obvious in the tillering stage, NIS had the highest NH4+-N content. The combination of rice straw and stabilized fertilizer delayed the peak of NH4+ release (Fig. 1). Thereafter, for rice cultivation, the NH4+ release character of NIS meets the rice demand, which is lower in seedling but higher in tillering stage (Chauhan et al. 2017). The delayed release of NH4+-N and rice high uptake in tillering coincide. This combination reduces the loss caused by large release and little demand, which means that there is a timing effect that is benefit for fertilizer utility and rice growth. While for NO3–-N content, the straw addition significantly reduced the NO3–-N content in the tillering stage and increased it in the maturation stage (P < 0.05), indicating the persistent release of NO3–-N with straw addition, which may increase the risk of gaseous release (N2O and NO) through de-nitrification (Xia et al. 2016). According to Ye and Horwath (2016), who concerned that water saturated conditions and high soil organic C (SOC) may lead to higher risk of N2O emission. Further, the lower NO3–-N in tillering stage occurred because of the immobilization of NH4+ by microbial biomass (Fig. 2). However, there were no significant influences of NO3–-N on stabilized fertilizer and normal urea.
Fig. 1. Soil NH4+-N and NO3–-N contents with straw and fertilizer applications. a: NH4+-N content; b: NO3–-N content. Values are the means of three; lowercase letter represents the significant difference between treatments during the same sampling period (Duncan, P < 0.05).
Effects of Stabilized Fertilizer and Straw on Soil Microbial Biomass C and N, and Dissolved Organic C and N
Soil dissolvable organic carbon (DOC) and microbial biomass carbon (MBC) were both effective soil carbon pools, which are variable components in soil and can be easily used by microorganisms in the process of transformation and mineralization. The addition of straw was expected to promote the microbial assimilations of C and N (Fig. 2), the soil contents of DOC, dissolvable organic nitrogen (DON), MBC, and microbial biomass nitrogen (MBN), which significantly increased at seedling and tillering stages (P < 0.05). Research shows that flooding conditions had a tendency to reduce C mineralization with a lower net residue-C mineralization. The mineralized C mainly comes from the recalcitrant pools, such as cellulose and lignin (Olivier and Horwath 2000). However, a higher SOC content effectively improved the crop nutrient supply and soil properties, increased the biodiversity, and enhanced the microbial activity (Yang et al. 2015). Similarly, straw addition stimulated soil respiration rate and total phospholipid fatty acids (Pan et al. 2016; Zhou et al. 2020). It also led to microbial assimilation of soil organic nitrogen, such as amino acids, to meet their need for C, N, and energy (Geisseler and Horwath 2014), and reduced the loss of urea and straw nitrogen. Meanwhile, the dead metabolism of microorganisms can release nitrogen for absorption and the subsequent utilization by crops in the later growth stage (Geisseler et al. 2011).
The inhibitor played an important part in the rice seedling and tillering stages. No significant effect was found during N and NI treatments. Compared with NS, stabilized urea with straw (NIS) reduced soil MBN and increased the DON content (Fig. 2). That discrepancy may cause the mineralized N in a later growth stage to meet the demand of rice, which may explain the highest N adsorption of UIS as shown in Table 2. A study by Geisseler et al. (2017) showed that fertilizer application could increase soil MBC and SOC contents. Except for delaying the hydrolysis of urea, the addition of inhibitors also increased the content of organic carbon (Fig. 2c), which was constant as reported by Wang et al. (1991). The combination of rice straw and inhibitor significantly influenced soil DOC and MBC (P < 0.05). Geisseler (2010) reported that the SOC positively correlated with the amount of cellulose added, which also showed a better N supply pattern. The higher SOC and improved soil nutrient status mainly attributed higher rice yield (Bi et al. 2009). Hence, the combination enhanced the turnover capacity of straw and the fixation effect of microorganisms on fertilizer nitrogen. The C-N coupling effect was found for straw decomposition and N acquisition.
Fig. 2. The content of dissolvable organic C (DOC) and N (DON) and soil microbial biomass C (MBC) and N (MBN) contents; a: DOC; b: DON; c: MBC; d: MBN. Uppercase letters represent the significant difference of treatments at different times of the same treatment, and lowercase letters represent the significance of different treatments at the same time (Duncan, p < 0.05).
Response of Stabilized Fertilizer and Straw on Soil Enzyme Activity
Activity of enzymes related to carbon conversion
Soil enzyme activities varied significantly with different managements of straw and stabilized fertilizer (P < 0.05), which also changed in various rice growth stages (Figs. 2 and 3). Straw addition significantly improved the activity of α-glucosidase, β-glucosidase, β-xylosidase, and β-cellulose compared to single urea application (P < 0.05, Fig. 3). However, the stabilized fertilizer inhibited the activities of α-glucosidase, β-xylosidase, and β-cellulose activities. Compared with NI, the AG enzyme activity of NS and NIS increased 32.4% and 56.4% in the seedling stage, 44.5% and 6.1% in the tillering stage, and 155.4% and 69.7% in the maturation stage, respectively (Fig. 3A). The BG enzyme was involved in the accumulation and conversion of organic carbon and played an important role in the decomposition of straw until maturation stage. The BG was dominated in NS and NI treatments at the seedling stage, while NS and NIS treatments dominated at the tillering stage (Fig. 3B). The variation of β-xylosidase (XYL) activity was consistent with α-glucosidase activity (Fig. 3A, Fig. 3C).
Fig. 3. Activity of α-D-glycosidase enzymes (MU-AG), β-D-glycosidase enzymes (MU-BG), β-D-xylosidase enzymes (MU-XYL), and β-D-cellulose (MU-CEL) enzymes related to the turnover of carbon in the soil. A: MU-AG; B: MU-BG; C: MU-XYL; D: MU-CEL
Compared with stabilized fertilizer, the NS and NIS treatments increased XYL activity by 291.6% and 348.5% in the seedling stage and 650.7% and 287.9% in the maturation stage, respectively, due to straw addition (Fig. 3C). According to Fig. 3D, NS had the highest β-cellulose (CEL) enzyme activity in the whole growth period; NS, NI, and NIS treatments increased the activity by 61.4%, 11.9%, and 23.9% at the seedling stage, and 58.8%, 16.3%, and 31.9% at the tillering stage, respectively, compared with urea alone. Qiu et al. (2011) reported that straw applied increased the organic matter, which is easily decomposed by small molecules, approximately 80% of cellulose of which is difficult to be decomposed. However, the products of cellulose degradation are glucose, cellobiose, and higher molecular weight oligosaccharides (Deng and Tabatabai 1994). Glucosidase and cellulase were the main participants in the hydrolysis and utilization of cellulose in the soil; the increase of the C-acquiring enzyme makes more C available to soil microorganisms (An et al. 2015). This explained why the decomposition of straw addition significantly improved the activity of hydrolysis enzymes compared to single urea application (P < 0.05). Moreover, with rice growth, the root system grows rapidly, the exudates and exfoliates of the root system increased, and the rich matrix promotes the increase of soil microorganisms and soil enzyme activity (Kader et al. 2017). However, there was no significant effect found in urea and stabilized fertilizer on soil carbon related hydrolytic enzymes under no straw addition.
Activity of enzymes related to nitrogen conversion
The index of soil N availability and the activity of N-acquiring enzyme such as N-acetyl-β-glucosaminidase (NA) and protease (PG) were measured, which was regulated by N availability. As shown in Fig. 4, the NA activity represented a trend of tillering stage > seedling stage > maturation stage. There was no significant effect on soil C-related hydrolytic enzymes of stabilized fertilizer (P > 0.05), but it showed an inhibitory effect on NA activity among the entire growing stage with or without straw. Meanwhile, PG showed adverse phenomena compared with NA enzyme.
Fig. 4. Activity of N-acetyl-β-glucosaminidase (NA) and protease (PG) enzymes related to N turnover in soil. a: MU-NA; b: PG.
The activity of PG reflects soil N conditions, protease hydrolysis produced polypeptide, and then hydrolyzed to amino acid. The hydrolysis product is an important source of N for plants and microbes, after macromolecular aggregation under the catalysis of PG absorbed by microorganisms (Yang et al. 2017). Results showed that straw addition (NS, NIS) significantly increased soil protease activity at the tillering and maturation stages. Stabilized fertilizer showed a noticeably higher protease activity, which means that the inhibitor promoted the PG metabolisms and the microorganisms still had higher activity, and N turnover capacity, mainly due to the availability of nitrogen substrates by microorganism’s absorption and utilization still be high compared with urea alone. Research showed that the nitrification inhibitor NBPT impeded the growth of ammonia-oxidizing archaea (AOA), and DMPP significantly reduced the ammonia oxidizing bacterial abundance, which significantly inhibited urea hydrolysis indirectly affecting nitrification (Dong et al. 2013; Meng et al. 2020). Geisseler et al. (2010) showed that the N turnover was determined by the amount and species of N source. This study showed that the domination of metabolism can be changed with added rice straw, which altered the C/N ratio of soil in the whole incubation period (Table 1). Yu et al. (2020) reported that straw and inhibitor influenced the fertilizer nitrogen transformation into soil organic pools. Rice straw significantly increased soil proteinase activity also (P<0.05, Fig 4). The present research found that stabilized fertilizer combined with straw may change the metabolic pathway of N, which increased protease activity but decreased NA activity, the specific bacteria needs further validation.
Principal Component Analysis Among Rice Yield, Soil Properties, and Soil Enzyme Activities
The principal component analysis (PCA) of enzyme activity with soil C and N is shown in Fig. 5.
The PCA produced two principal components. The first and the second principal components explained 76.60% of the total variability of the results. Results showed that the addition of straw significantly affected soil DOC, DON, MBC, and MBN (P < 0.05); the enzymes related to carbon conversion were closely related to the content of organic nitrogen in soil. Wang (2019) verified that the type of soil management employed affected microbial biomass and soil enzyme activities. Enzyme activities strongly depended on microbial activity and biomass, as microorganisms produce enzymes depending on resource supply and demand (Mooshammer et al. 2014). Soil DOC provides direct organic substrates for microbial growth and reproduction, and the increase of substrate carbon source improved microbial activity and caused the increase of MBN content (Qiu et al. 2011; Liang et al. 2019). Sampling times influenced the enzyme activities and soil carbon transformation, especially in the tillering stage, which had a strong effect on it. And there are similar separations among different treatments based on the enzyme activities in seedling stage and maturation stage (Fig.5). The results were also similar with Zhou et al. (2020).
To better understand the effect of straw and stabilized urea on soil properties and enzyme activities, Spearman’s correlation analysis was performed. According to Table 3, soil BG, NA, and PG enzymes and DOC, DON, NH4+-N, and NO3–-N contents played an important role on rice yield. There was a negative correlation between the inhibitor and C-acquiring enzymes (AG, BG, XYL, and CEL), but the inhibitor could significantly increase the NA activity (P < 0.05, Table 3).
Geisseler (2014) reported that nitrogen reduced C-acquiring enzymes. The same effect was found of stabilized fertilizer in this experiment. Straw addition was positively related with CEL and XYL activities and soil DOC and MBN contents. Zhang (2014) also considered that carbon material addition was closely related to microbial decomposition characteristics under fertilization conditions. Especially under the condition of flooding and hypoxia, the activity of hydrolytic enzyme is inhibited, leading to a large amount of accumulation of organic matter (Freeman et al. 2001; Dunn and Freeman 2018). Moreover, stabilized fertilizer combined with straw may change the metabolic pathway of N. This may be explained by PG that had a significant positive correlation with NH4+-N, while NA was positively related to NH4+-N, NO3–-N and negatively related to the inhibitor addition according to Table 3. Thus, there may be a difference between the metabolic processes used by the microorganisms.
Note: Duncan method was used for correlation analysis, with * P < 0.05 and ** P < 0.01
Fig. 5. PCA analysis on enzyme activity (α-D-glycosidase enzymes, AG; β-D-glycosidase enzymes, MU-BG); N-acetyl-β-glucosaminide enzymes, MU-NA; β-D-xyloside, MU-XYL; β-D-cellobioside enzyme, MU-CEL; Protease enzyme, PG) related to soil carbon nitrogen (DOC, DON, MBC, and MBN) in paddy soil (n = 9). The letter before the treatment abbreviations: S represents seedling stage, T represents tillering stage, and M represents maturation stage.
CONCLUSIONS
- Rice straw significantly increased the dissolved carbon/nitrogen and microbial biomass carbon/nitrogen at the seedling and tillering stages, and it increased enzyme activity related to carbon turnover.
- Stabilized fertilizer made of urea with urease inhibitor 1%PPD + 1%NBPT and nitrification inhibitor 2% DMPP enhanced N use efficiency, changed the metabolic pathway of N, mainly due to PG activity closely associated with NH4+-N compared with NA enzyme, and increased rice productivity.
- There was a win-win effect of straw and stabilized fertilizer on soil organic carbon accumulation and crop productivity through increasing N efficiency by enhancing microbial activity. Rice straw coupled with the new type of stabilized fertilizer is a promising agricultural management strategy among paddy soils in north China.
ACKNOWLEDGMENTS
The authors are grateful for the K. C. Wong Education Foundation, National Scientific Foundation Project of China (31971531; 41807388), and the financial support of the State Key Program of China (2016YFD0300904; 2017YFD020070). The authors are grateful to the Shenyang Agricultural University Rice Research Institute for providing the testing soil and the National Field Research Station of Shenyang Agro-ecosystems, Chinese Academy of Sciences, for providing site to conduct the experiment.
REFERENCES CITED
An, T., Schaeffer, S., Zhuang, J., Radosevich, M., Li, S., Li, H., Pei, J., and Wang, J. (2015). “Dynamics and distribution of 13C-labeled straw carbon by microorganisms as affected by soil fertility levels in the Black Soil region of Northeast China,” Biology and Fertility of Soils 51(5), 605-613. DOI: 10.1007/s00374-015-1006-3
Bi, L. D., Zhang, B., Liu, G. R., Li, Z. Z., Liu, Y. R., Ye, C., Yu, X. C., Lai, T., Zhang, J. G., Yin, J. M., et al. (2009). “Long-term effects of organic amendments on the rice yields for double rice cropping systems in subtropical China,” Agriculture Ecosystems & Environment 129(4), 534-541. DOI: 10.1016/j.agee.2008.11.007
Bowles, T. M., Acosta-Martínez, V., Calderón, F., and Jackson, L. E. (2013). “Soil enzyme activities, microbial communities, and carbon and nitrogen availability in organic agroecosystems across an intensively-managed agricultural landscape,” Soil Biology and Biochemistry 68, 252-262. DOI: 10.1016/j.soilbio.2013.10.004
Chauhan, B. S., Jabran, K., and Mahajan, G. (2017). “Fertilizer management in rice,” in: Rice Production Worldwide. Springer, Gewerbestrasse, Switzerland, pp. 38-52. DOI 10.1007/978-3-319-47516-5
Chen, Z. M., Wang, H. Y., Liu, X. W., Zhao, X. L., Lu, D. J., Zhou, J. M., and Li, C. Z. (2017). “Changes in soil microbial community and organic carbon fractions under short-term straw return in a rice–wheat cropping system,” Soil and Tillage Research 165, 121-127. DOI: 10.1016/j.still.2016.07.018
Cucu, M. A., Said-Pullicino, D., Maurino, V., Bonifacio, E., Romani, M., and Celi, L. (2013). “Influence of redox conditions and rice straw incorporation on nitrogen availability in fertilized paddy soils,” Biology and Fertility of Soils 50(5), 755-764. DOI: 10.1007/s00374-013-0893-4
Deng, S. P., and Tabatabai, M. A. (1994). “Cellulase activity of soils,” Soil Biology and Biochemistry 26(10), 1347-1354. DOI: 10.1016/0038-0717(94)90216-X
Dong, X. X., Zhang, L. L., Wu, Z. J., Zhang, H. W., and Gong, P. (2013). “The response of nitrifier, N-fixer and denitrifier gene copy numbers to the nitrification inhibitor 3,4-dimethylpyrazole phosphate,” Plant Soil Environ. 59, 398-403. DOI: 10.17221/165/2013-PSE
Dunn, C., and Freeman, C. (2018). “The role of molecular weight in the enzyme-inhibiting effect of phenolics: The significance in peat land carbon sequestration,” Ecological Engineering 114, 162-166. DOI: 10.1016/j.ecoleng.2017.06.036
Eagle, A. J., Bird, J. A., Horwath, W. R., Linquist, B. A., Brouder, S. M., Hill, J. E., and van Kessel, C. (2000). “Rice yield and nitrogen utilization efficiency under alternative straw management practices,” Agronomy Journal 92(6), 1096-1103. DOI: 10.2134/agronj2000.9261096x
Freeman, C., Ostle, N., and Kang, H. (2001). “An enzymic ‘latch’ on a global carbon store – A shortage of oxygen locks up carbon in peatlands by restraining a single enzyme,” Nature 409(6817), 149-149. DOI: 10.1038/35051650
Fujita, K., Kunito, T., Matsushita, J., Nakamura, K., Moro, H., Yoshida, S., Toda, H., Otsuka, S., and Nagaoka, K. (2018). “Nitrogen supply rate regulates microbial resource allocation for synthesis of nitrogen-acquiring enzymes,” PLoS One 13(8), e0202086. DOI: 10.1371/journal.pone.0202086
Geisseler, D., and Horwath, W. R. (2014). “Investigating amino acid utilization by soil microorganisms using compound specific stable isotope analysis,” Soil Biology and Biochemistry 74, 100-105. DOI: 10.1016/j.soilbio.2014.02.024
Geisseler, D., and Scow, K. M. (2014). “Long-term effects of mineral fertilizers on soil microorganisms – A review,” Soil Biology and Biochemistry 75, 54-63. DOI: 10.1016/j.soilbio.2014.03.023
Geisseler, D., Linquist, B. A., and Lazicki, P. A. (2017). “Effect of fertilization on soil microorganisms in paddy rice systems – A meta-analysis,” Soil Biology and Biochemistry 115, 452-460. DOI: 10.1016/j.soilbio.2017.09.018
Geisseler, D., Horwath, W. R., and Scow, K. M. (2011). “Soil moisture and plant residue addition interact in their effect on extracellular enzyme activity,” Pedobiologia 54(2), 71-78. DOI: 10.1016/j.pedobi.2010.10.001
Geisseler, D., Horwath, W. R., Joergensen, R. G., and Ludwig, B. (2010). “Pathways of nitrogen utilization by soil microorganisms – A review,” Soil Biology and Biochemistry 42(12), 2058-2067. DOI: 10.1016/j.soilbio.2010.08.021
Inselsbacher, E., Hinko-Najera, Umana, N., Stange, F. C., Gorfer, M., Schüller, E., Ripka, K., Zechmeister-Boltenstern, S., Hood-Novotny, R., Strauss, J., et al. (2010). “Short-term competition between crop plants and soil microbes for inorganic N fertilizer,” Soil Biology and Biochemistry 42(2), 360-372. DOI: 10.1016/j.soilbio.2009.11.019
Jian, S., Li, J., Chen, J., Wang, G., Mayes, M. A., Dzantor, K. E., Hui, D., and Luo, Y. (2016). “Soil extracellular enzyme activities, soil carbon and nitrogen storage under nitrogen fertilization: A meta-analysis,” Soil Biology and Biochemistry 101, 32-43. DOI: 10.1016/j.soilbio.2016.07.003
Joergensen, R. G. (1996). “The fumigation-extraction method to estimate soil microbial biomass_ calibration of the kEC value,” Soil Biology and Biochemistry 28(1), 25-31. DOI: 10.1016/0038-0717(95)00102-6
Joergensen, R. and Mueller, T. (1996) “The fumigation-extraction method to estimate soil microbial biomass: Calibration of the KEN value,” Soil Biology and Biochemistry 28(1), 33-37. DOI: 10.1016/0038-0717(95)00101-8
Kader, M. A., Yeasmin, S., Solaiman, Z. M., De Neve, S., and Sleutel, S. (2017). “Response of hydrolytic enzyme activities and nitrogen mineralization to fertilizer and organic matter application in subtropical paddy soils,” European Journal of Soil Biology 80, 27-34. DOI: 10.1016/j.ejsobi.2017.03.004
Kandeler, E., Palli, S., Stemmer, M., and Gerzabek, M. H. (1999). “Tillage changes microbial biomass and enzyme activities in particle-size fractions of a Haplic Chernozem,” Soil Biology and Biochemistry 31(9), 1253-1264. DOI: 10.1016/s0038-0717(99)00041-3
Ladd, J. N., and Butler, J. H. A. (1972). “Short-term assays of soil proteolytic enzyme activities using proteins and dipeptide derivatives as substrates,” Soil Biology and Biochemistry 4(1), 19–30. DOI: 10.1016/0038-0717(72)90038-7
Liang, H., Hu, K., Qin, W., Zuo, Q., Guo, L., Tao, Y., and Lin, S. (2019). “Ground cover rice production system reduces water consumption and nitrogen loss and increases water and nitrogen use efficiencies,” Field Crops Research 233, 70-79. DOI: 10.1016/j.fcr.2019.01.003
Lu, C., Chen, H., Teng, Z., Yuan, L., Ma, J., He, H., Chen, X., Zhang, X., and Shi, Y. (2018). “Effects of N fertilization and maize straw on the dynamics of soil organic N and amino acid N derived from fertilizer N as indicated by 15N labeling,” Geoderma 321, 118–126. DOI: 10.1016/j.geoderma.2018.02.014
Lu, P., Zhang, Y., Jia, C., Wang, C., Li, X., and Zhang, M. (2015). “Polyurethane from Liquefied wheat straw as coating material for controlled release fertilizers,” BioResources 10(4), 7877-7888. DOI: 10.15376/biores.10.4.7877-7888
Ma, Q., Wu, Z., Shen, S., Zhou, H., Jiang, C., Xu, Y., Liu, R., and Yu, W. (2015). “Responses of biotic and abiotic effects on conservation and supply of fertilizer N to inhibitors and glucose inputs,” Soil Biology and Biochemistry 89, 72-81. DOI: 10.1016/j.soilbio.2015.06.024
Marx, M. C., Wood, M., and Jarvis, S. C. (2001). “A microplate fluorimetric assay for the study of enzyme diversity in soils,” Soil Biology and Biochemistry 33(12-13), 1633-1640. DOI: 10.1016/S0038-0717(01)00079-7
Meng, X. T., Li, Y. Y., Yao, H. Y., Wang, J., Dai, F., Wu, Y. P., and Chapman, S. (2020). “Nitrification and urease inhibitors improve rice nitrogen uptake and prevent denitrification in alkaline paddy soil,” Applied Soil Ecology 154, 103665. DOI: 10.1016/j.apsoil.2020.103665
Mooshammer, M., Wanek, W., Hammerle, I., Fuchslueger, L., Hofhansl, F., Knoltsch, A., Schnecker, J., Takriti, M., Watzka, M., Wild, B., et al. (2014). “Adjustment of microbial nitrogen use efficiency to carbon:nitrogen imbalances regulates soil nitrogen cycling,” Nature Communication 5, 3694. DOI: 10.1038/ncomms4694
Olivier, C. D., and Horwath, W. R. (2000). “Decomposition of rice straw and microbial carbon use efficiency under different soil temperatures and moistures,” Soil Biology and Biochemistry 32, 1773-1785. DOI: S0038- 0717(00)00096-1
Pan, F., Li, Y., Chapman, S. J., Khan, S., and Yao, H. (2016). “Microbial utilization of rice straw and its derived biochar in a paddy soil,” Science of The Total Environment 559, 15-23. DOI: 10.1016/j.scitotenv.2016.03.122
Qiu, S. J., Peng, P. Q., Li, L., He, P., Liu, Q., Wu, J. S., Christie, P., and Ju, X. T. (2011). “Effects of applied urea and straw on various nitrogen fractions in two Chinese paddy soils with differing clay mineralogy,” Biology and Fertility of Soils 48(2), 161-172. DOI: 10.1007/s00374-011-0613-x
San Francisco, S., Urrutia, O., Martin, V., Peristeropoulos, A., and Garcia-Mina, J. M. (2011). “Efficiency of urease and nitrification inhibitors in reducing ammonia volatilization from diverse nitrogen fertilizers applied to different soil types and wheat straw mulching,” Journal of the Science of Food and Agriculture 91(9), 1569-1575. DOI: 10.1002/jsfa.4349
Shi, Y., Zhang, L., and Zhao, M. (2015). “Effect of biochar application on the efficacy of the nitrification inhibitor Dicyandiamide in soils,” BioResources 10(1), 1330-1345. DOI: 10.15376/biores.10.1.1330-1345
Silva, A. G. B., Sequeira, C. H., Sermarini, R. A., and Otto, R. (2017). “Urease inhibitor NBPT on ammonia volatilization and crop productivity: A meta-analysis,” Agronomy Journal 109(1), 1-13. DOI: 10.2134/agronj2016.04.0200
Wang, Z., Van, O., Cleemput, Li, L., and Baer, L. (1991). “Effect of organic matter and urease inhibitors on urea hydrolysis and immobilization of urea nitrogen in an alkaline soil,” Biology and Fertility of Soils 11(2), 101-104. DOI: 10.1007/bf00336372
Wang, Q., Ma, M., Jiang, X., Guan, D., Wei, D., Zhao, B., Chen, S., Cao, F., Li, L., Yang, X. et al. (2019). “Impact of 36 years of nitrogen fertilization on microbial community composition and soil carbon cycling-related enzyme activities in rhizospheres and bulk soils in northeast China,” Applied Soil Ecology 136, 148-157. DOI: 10.1016/j.apsoil.2018.12.019
Wu, K. K., Gong, P., Zhang, L. L., Wu, Z. J., Xie, X. S., Yang, H. Z., Li, W. T., Song, Y. C., and Li, D. P. (2019) “Yield-scaled N2O and CH4 emissions as affected by combined application of stabilized nitrogen fertilizer and pig manure in rice fields,” Plant, Soil and Environment, 65, 497-502. DOI: 10.17221/286/2019-pse
Xia, L., Xia, Y., Ma, S., Wang, J., Wang, S., Zhou, W., and Yan, X. (2016). “Greenhouse gas emissions and reactive nitrogen releases from rice production with simultaneous incorporation of wheat straw and nitrogen fertilizer,” Biogeosciences 13(15), 4569-4579. DOI: 10.5194/bg-13-4569-2016
Yang, H., Yang, B., Dai, Y., Xu, M., Koide, R. T., Wang, X., Liu, J., and Bian, X. (2015). “Soil nitrogen retention is increased by ditch-buried straw return in a rice-wheat rotation system,” European Journal of Agronomy 69, 52-58. DOI: 10.1016/j.eja.2015.05.005
Yang, L., Zhang, L., Yu, C., Li, D., Gong, P., Xue, Y., Song, Y., Cui, Y., Doane, T. A., and Wu, Z. (2017). “Nitrogen fertilizer and straw applications affect uptake of 13C, 15N-glycine by soil microorganisms in wheat growth stages,” PLoS One 12(1), e0169016. DOI: 10.1371/journal.pone.0169016
Yang, L., Zhang, L., Geisseler, D., Wu, Z., Gong, P., Xue, Y., Yu, C., Juan, Y., and Horwath, W. R. (2016). “Available C and N affect the utilization of glycine by soil microorganisms,” Geoderma 283, 32-38. DOI: 10.1016/j.geoderma.2016.07.022
Ye, R., and Horwath, W. R. (2016). “Nitrous oxide uptake in rewetted wetlands with contrasting soil organic carbon contents,” Soil Biology and Biochemistry 100, 110-117. DOI: 10.1016/j.soilbio.2016.06.009
Zhang, Y. L., Chen, L. J., Chen, X. H., Tan, M. L., Duan, Z. H., Wu, Z. J., Li, X. J., and Fan, X. H. (2015). “Response of soil enzyme activity to long-term restoration of desertified land,” CATENA 133, 64-70. DOI: 10.1016/j.catena.2015.04.012
Zhou, G., Gao, S., Lu, Y., Liao, Y., Nie, J., and Cao, W. (2020). “Co-incorporation of green manure and rice straw improves rice production, soil chemical, biochemical and microbiological properties in a typical paddy field in southern China,” Soil and Tillage Research 197, Article ID:104499. DOI: 10.1016/j.still.2019.104499
Article submitted: November 13, 2020; Peer review completed: February 13, 2021; Revised version received and accepted: March 2, 2021; Published: March 5, 2021.
DOI: 10.15376/biores.16.2.3059-3074
