Abstract
Teakwood from fast-growth plantations is commercialized at increasingly younger ages for economic reasons. However, wood features are influenced by the age of the tree. This study examined how age affects heartwood proportion, color parameters, chemical composition, and natural teakwood durability. Trees with 5, 10, 15, and 20 years of fast-growth at commercial plantations located in Mato Grosso, Brazil were evaluated. The base diameter of the trees ranged from 13.1 (5 years) to 28.3 cm (20 years), and the heartwood percentage increased from 16.3 to 60.0%, respectively. The color parameters in the CIELab system indicated that wood became darker and more saturated, and the predominance of yellow color decreased compared with red as age advanced. The total extractive content ranged between 7.4 (5 years) and 9.6% (15 years), without a clear trend of age affecting the extractive content. The extractives from five-year-old wood were mainly composed of tectoquinone (43.3%), phthalic acid (19.1%), and 1,3-indandione (9.2%), while those from 20-year-old wood were mainly composed of tectoquinone (60.7%), lapachol (13.8%), and phthalic acid (9.7%). Teakwood can be classified as resistant (5 years) to very resistant (20 years) after being submitted to an accelerated decay test.
Download PDF
Full Article
Effect of Age on Heartwood Proportion, Color, Chemical Composition, and Biological Resistance of Teakwood
Gabriel Afonso de Oliveira Silva,a Kezia Rage Curvo,a Aylson Costa Oliveira,a Pedro Nicó Medeiros Neto,b Leonardo Gomes Vasconcelos,a Ana Márcia Macedo Ladeira Carvalho,c Márcio José Silva,c Ricardo Natalino,c and Bárbara Luísa Corradi Pereira a,*
Teakwood from fast-growth plantations is commercialized at increasingly younger ages for economic reasons. However, wood features are influenced by the age of the tree. This study examined how age affects heartwood proportion, color parameters, chemical composition, and natural teakwood durability. Trees with 5, 10, 15, and 20 years of fast-growth at commercial plantations located in Mato Grosso, Brazil were evaluated. The base diameter of the trees ranged from 13.1 (5 years) to 28.3 cm (20 years), and the heartwood percentage increased from 16.3 to 60.0%, respectively. The color parameters in the CIELab system indicated that wood became darker and more saturated, and the predominance of yellow color decreased compared with red as age advanced. The total extractive content ranged between 7.4 (5 years) and 9.6% (15 years), without a clear trend of age affecting the extractive content. The extractives from five-year-old wood were mainly composed of tectoquinone (43.3%), phthalic acid (19.1%), and 1,3-indandione (9.2%), while those from 20-year-old wood were mainly composed of tectoquinone (60.7%), lapachol (13.8%), and phthalic acid (9.7%). Teakwood can be classified as resistant (5 years) to very resistant (20 years) after being submitted to an accelerated decay test.
DOI: 10.15376/biores.18.2.4116-4131
Keywords: Tectona grandis; CIELab; GC-MS; Trametes versicolor
Contact information: a: Federal University of Mato Grosso, Cuiabá, MT, Brazil; b: Federal University of Campina Grande, Campina Grande, PB, Brazil; c: Federal University of Viçosa, Viçosa, MG, Brazil;
* Corresponding author: barbara.pereira@ufmt.br
GRAPHICAL ABSTRACT

INTRODUCTION
The species Tectona grandis L.f., Lamiaceae, known as teak, produces one of the most valuable tropical woods in the global market. However, the characteristics that make it valuable in luxury markets, such as yacht and furniture manufacturing, and in construction industry, are related to the wood from natural forests in the Asian continent. This wood presents a set of features, in which the average density is associated with very high durability against biological agents, high dimensional stability, and attractive aesthetic characteristics, mainly related to the color of the heartwood and the growth ring patterns (Kollert and Kleine 2017).
Natural teak forests have reduced significantly in the past century due to overexploitation and land use conversion into plantations and pastures (Ugalde Arias 2013). In this context, the wood market for this plantation-derived species has become promising. The species is grown in 70 countries in Asia, Africa, Latin America, and Oceania, and the forests planted with the species are estimated to occupy 6.89 million hectares (Kollert and Kleine 2017). In the American continent, the largest producers are Panama, Costa Rica, Equador, and Brazil (Camino and Morales 2013).
In 2018, Brazil had 93,957 hectares planted with teak (IBÁ, 2021). Besides teak’s rapid growth in some Brazilian areas, the formation of straight trunks has encouraged the large-scale planting of the species for sawn timber production for export (Leite et al. 2011). Most of the teak plantations in the country are established with private investments and are therefore managed in short rotations, with final harvesting between 20 and 25 years (Ugalde Arias 2013). However, according to the author, due to the high costs of establishment and maintenance, the tendency is that the final harvest age does not exceed 20 years. As silvicultural practices, thinning and pruning are adopted to regulate competition between trees and produce clean, knot-free wood. The first thinning is usually done from 3 to 6 years, and there are subsequent ones, up to a total of four, with the last one at 15 years of age. The sale of wood from thinning generates an intermediate revenue, which helps to cover the initial establishment and maintenance costs.
The value of the teakwood from fast-growth plantations in Brazil can reach US$600.00 per hoppus cubic meter (ITTO 2022), for logs with diameters larger than 35 cm for wood harvested at the end of the rotation cycle. In contrast, wood derived from the first thinning, normally at five years old, is sold to the local market to be used as firewood for around US$ 5.00 per cubic meter, which does not pay for the costs of harvest, removal, and transport of the wood.
It is worth highlighting that the valuation of teakwood is especially associated with log dimensions – both diameter and length – and heartwood proportion, and the age of the tree will influence the value of the wood. The heartwood proportion decreases along the longitudinal position of the tree. That is, the logs of older trees that are closer to the base have a larger heartwood proportion. In addition, the heartwood percentage rises with the growth rate of the trees; therefore, logs of large diameters can be produced with larger heartwood proportion in short-rotation plantations by selecting superior genetic materials and accelerating the growth of the trees using forestry practices (Ugalde Arias 2013).
The natural durability of teak increases with age and heartwood proportion due to the higher concentration of toxic phenolic extractive through the action of xylophagous organisms present in the heartwood (Siqueira et al. 2020). Furthermore, the presence of extractives in the wood influences not only its durability but also its color, which can be attributed to the presence, amount, and type of extractive (Brocco et al. 2017). There is a correlation between color and natural resistance of wood, which, in turn, is related to the species, amount, and type of extractives present (Amusant et al. 2004). In addition, the factor of color has been analyzed as potentially affecting the price of wood (Ribeiro et al. 2017).
This study investigated the effect of teakwood age on the production of heartwood, color parameters, chemical composition, and natural durability. It is an important tool for the valuation of teak lumber from short-rotation plantations and the use of younger wood derived from thinning.
EXPERIMENTAL
Material
The teakwood (Tectona grandis Linn. F) came from commercial plantations belonging to the company Guavirá Florestal and Agroindustrial Ltda, in the city of Nova Maringá, Mato Grosso, Brazil (Latitude: 13° 1′ 2” South, Longitude: 57° 4′ 8” West) (Table 1). It is in the Cerrado biome, whose climate is characterized as tropical with a dry season (Köppen-Geiger climate classification: Aw), and mean annual temperature and rainfall reaches 24 °C and 1600 mm, respectively. The altitude varies between 450 and 500 m, and the predominant soil class is Red Yellow Latosol.
Table 1. General Information on the Teak Plantations in Nova Maringá, MT, Brazil
Caption: DBH = Diameter at breast height with bark.
Twelve trees of medium diameter were randomly sampled for each material studied, totaling 48 trees. The selection was made by excluding trees that did not have good phytosanitary conditions and those located on the edges of the plantations. A 5 cm thick disk was taken at the base of each tree (0.3 m), which was used as material in this study. The twelve trees per treatment were evaluated in the dendrometric and colorimetric analyses. For the chemical analyses and biodeterioration test, six trees per treatment were used.
Diameter and Proportion of Heartwood
The discs were subjected to natural drying at a mean temperature of 25 °C and mean relative humidity of 65% for 30 days, and subsequently they were polished on a belt grinder with grit 100. Then, the discs were photographed, and their barks, heartwoods, and sapwoods were distinguished based on their color differences, with the areas delimited and measured through ImageJ software (Schneider et al. 2012), with a calibration of 1.0 mm. The mean diameters without bark and the heartwood of each disc were also measured. The heartwood percentage was obtained by the ratio of the heartwood area and the disc area without bark, multiplied by 100.
Color Parameters
The color parameters were measured by the CIEL*a*b* system using a portable spectrophotometer (CR-410, Konica Minolta, Osaka, Japan). Two perpendicular lines were traced on the discs passing through the pith and two readings were performed per disc, at the cross-section, on two opposite sides. The color parameters of luminosity (L*), green-red axis (a*), blue-yellow axis (b*), chroma (C*), and hue angle (hº) were determined. The equipment configurations corresponded to a 5-mm sensor opening, a D65 light source (illuminant) containing a xenon bulb, a 10º standard supplementary observer, and an illumination area of 53 mm in diameter.
Elemental and Chemical Composition
The discs were divided into four wedge-shaped parts that passed through the pith, resulting in one sample per tree comprising a combination of heartwood and sapwood. Two opposite wedges were cut into wood strips, which were then milled using a Willey-type mill (MA304, Marconi, Piracicaba, Brazil). The resulting sawdust was classified using sieves with 40 mesh (0.40 mm) and 60 mesh (0.25 mm). Elemental and chemical analyses were performed on each tree in duplicate.
The elemental analysis was performed using the CHNS equipment (TruSpec Micro, Leco, St. Joseph, MI, USA), and the percentual of carbon (C), hydrogen (H), nitrogen (N), and sulfur (S) were determined in the TruSpur CHNS Micro module. The percentage of oxygen (O) was obtained by the sum of percentages of C, H, N, and ashes subtracted from 100. The content of ashes was determined according to D1102-84 (ASTM, 2013).
The total extractive content was determined according to the methodologies described in ASTM D1105-96 (2013) through successive extractions in ethanol-toluene (2:1) and in ethanol, in Soxhlet device, followed by a hot water bath. The content of insoluble lignin was determined following the Klason method, modified according to the procedure proposed by Gomide and Demuner (1986). The remaining filtrate from the Klason lignin analysis allowed for a spectrophotometer reading to determine the soluble lignin in acid, according to Goldschmid (1971). The total lignin content was obtained by the sum of the contents of Klason and soluble lignins. The holocellulose content was determined by adding the total lignin, total extractives, and ash contents subtracted from 100.
GC-MS Analysis
Trees aged 5 and 20 years were selected for comparison of the teakwood extractives obtained during the first thinning and at the final harvest age. A 100 g sample was prepared for each age, using approximately 17 grams of heart-sapwood sawdust from each of the six trees per treatment. Consecutive extractions were performed using ethanol-toluene (2:1), ethanol, and hot water, following the methodology mentioned above. Subsequently, the extracts obtained for each treatment were concentrated using a rotaevaporator.
An aliquot of the extracts was previously diluted in hexane (50 μL of sample for 1 mL of hexane) for analysis of gas chromatography coupled to mass spectrometry (GC-MS). The GC analysis was performed on Shimadzu equipment (GC-2010 Plus, Shimadzu, Kyoto, Japan). A capillary column RTX – 5 (30 m in length × 0.250 mm in internal diameter x 0.25 μm in thickness) was used, at a temperature of 40 °C to 240 °C at 3.5 °C per min. Both the injector and detector were kept at 250 °C, and helium was used as a gas carrier at a pressure of 0.04 MPa with an input flow rate of 3 mL.min-1 and a total flow of 46.2 mL.min-1. A volume of 0.4 μL of the samples using a Split of 1:100 was injected. The MS analyses were performed using Shimadzu equipment (GCMS – QP2010 Ultra, Shimadzu, Kyoto, Japan) operating at 70 eV, and the ion source temperature was kept at 200 °C. The components were identified from the fragmentation of their masses by comparing the mass spectra of the components with the spectra of the reference libraries Adams (2007) and Nist (1990), stored in the EM database. The retention time in the column and the percentage of each chemical compound identified were also obtained.
Biodeterioration of Teak Wood by Rot Fungus
Similarly to the previous analysis, the biodeterioration test compared the ages of 5 and 20 years. Radial wood samples measuring 2.5 cm in width and 1.0 cm in thickness were collected from the discs at the base of each tree, oriented from the pith to the bark. Each sample was then tangentially divided into parts measuring approximately 2.5 cm. For the 5-year-old trees, three test specimens were obtained per tree, based on a mean radius of 8.0 cm. For the 20-year-old trees, with a mean radius of 18.0 cm, seven test specimens were obtained per tree.
The loss of mass and the resistance classes resulting from the fungal action after 16 weeks were estimated following the methodology described in ASTM D-2017 (2005). A xylophagous fungus was used, Trametes versicolor (Linnaeus ex Fries) Pilat, derived from the Mad 697 strain (Culture Collection Center for Forest Mycology Research– CFMR/USA). The materials used were bottles of 200 mL with a screw cap containing sterile soil, 20 mL of distilled water, and a feeder strip of susceptible wood, Jacaranda copaia (Aubl.) D.Don. The resistance classes were also determined according to the classification based on the loss of mass: very resistant (0-10%); resistant (11-24%); moderate resistance (25-44%), and nonresistant (≥45%), according to D-2017 (ASTM 2005). As a reference, 15 test specimens of J. copaia wood (2.5 x 2.5 x 1.0 cm) were used. The reference test specimens indicate the vegetative vigor of the fungal culture used and the incubation conditions. The test was considered valid only if the fungus caused a loss of over 50% of mass in the reference species within 16 weeks (ASTM 2005).
Statistical Analysis
A Completely Randomized Design was used: four treatments (5, 10, 15, and 20 years) and a variable number of replications (trees): 12 for dendrometric and colorimetric analyses and six for chemical composition. In the biodeterioration assay, the wood mass loss was compared in three and seven radial positions, from pith to bark, at the ages of 5 and 20 years.
The data generated were analyzed by observing normality through the Shapiro-Wilk test and homogeneity of variance through the Bartlett test, both assessed (p-value<0.05). All data were then subjected to an analysis of variance. If any significant differences appeared, the means of each treatment were compared through the Tukey test (p-value<0.05). RStudio (R Corel Team, Vienna, Austria) was used for all data analysis.
RESULTS AND DISCUSSION
Diameter and Proportion of Heartwood
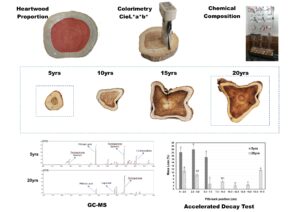
The photographs shown in Table 2 illustrate that as the age of teak trees advances, tabular roots or buttresses are formed at their bases.
Table 2. Image of the Discs and Dendrometric Variables Measured at the Base of Trees Teak at Different Ages
Values in brackets correspond to the standard deviation of the mean and lower-case letters in each line indicate significant differences according to the Tukey test (p-value<0.05).
The discs are round only for the age of 5 years, while the others have an irregular shape (Table 2). Such an irregularity at the base of these trees is related to the hormonal mechanisms that control the formation of wood over the years (Gaitan-Alvarez et al. 2019), and tabular roots help to anchor the tree to the ground and provide stability. But the irregularity at the base of the trees also has implications for their utilization in the timber industry. They affect the use of logs from the base, making it necessary to cut taller trees for logging (Takizawa 2022). Therefore, genetic improvement programs should take into account the shape of the tree base when selecting individuals for sawn wood production.
As observed in Table 2, the regions of heartwood and sapwood were differentiated by the change in color of the discs on the base of the trees at all ages assessed. The contrast between the golden-brown heartwood and the light-yellow sapwood makes it easy to distinguish between the two. The formation of heartwood in teak trees begins when the diameter ranges between 6 and 10 cm (Moya et al. 2014). The mean diameter of the five-year-old (13.1 cm) indicated that the formation of heartwood had already started.
As shown in Table 2, the diameter at the base of teak trees increases with age, with a double increase when comparing the ages of 5 and 15 years. Moreover, it was found that the base logs only reach diameters larger than 25 cm by the age of 15 years, which is better accepted in the international market. This is an important finding because exporting logs 15-25 cm in diameter, like base logs of 10-15-year-old trees, is often not feasible because of the low market value and logistic issues (Takizawa 2022). Thus, for value-adding, logs derived from thinning operations, with between 15 and 25 cm in diameter, are used by the wood industry for producing lumber and edge-glued panels. On the other hand, logs with diameters below 15 cm, logs with diameters below 15 cm, such as base logs of 5-year-old trees, are destined for the domestic market for energy production as firewood or chip, or even the production of fence posts (Takizawa 2022). This information is essential for forest managers and the timber industry, as it enables them to make informed decisions regarding the timing of thinnings and the final harvest. By taking into account the growth patterns and commercial value of the different age classes, they can optimize the management of teak plantations and increase profitability.
As to heartwood, where the commercial value of teakwood lies, the diameter and percentage of heartwood increase with age, with the largest increases occurring in the older materials (Table 2). The heartwood diameter increased 142% from five to ten years, while the average diameter increased by 56%. According to Pereira et al. (2013), the heartwood of the trees develops as their age advances; that is, the heartwood proportion is expected to increase as the tree ages. It is also worth highlighting another feature that differentiates teakwood from other forest species in the international market, namely the exponential growth of heartwood at a young age, which adds to its commercial value (Sanquetta et al. 2014).
When comparing the ages of 15 and 20 years, there were non-significant increases of 7% in the diameter of the tree base and 9% in the heartwood diameter. This fact, combined with the trees at 15 years already reaching a diameter of 25 cm, may justify the proposed reduction of the final harvest to ages below 20 years, as suggested by Ugalde Arias (2013).
Color Parameters
The age has a significant effect on teakwood color, as indicated by the F test which showed a significant difference (p-value< 0.05) for all color parameters (Table 3). The 5- and 10-year-old woods were lighter, while the 15- and 20-year-old woods were darker. Despite all the assessed woods being classified as light according to the classification by Camargos and Gonçalez (2001), the wood darkens with age. The quinone group, which is concentrated especially in the heartwood region, has been identified as a major contributor to wood darkening (Mehats et al. 2021; Qiu et al. 2019).
Table 3. Color Parameters in the CIELab System for Teakwood at Different Ages
Caption: L (luminosity); a* (green-red axis); b* (blue-yellow axis); C (Chroma); h* (hue angle). Values in brackets correspond to standard deviation and lower-case letters in each column indicate significant differences according to the Tukey test (p-value<0.05).
The study revealed that a* values ranged from 3.4 to 5.4, while b* ranged from 13.1 to 16.0, indicating a predominance of yellow pigments instead of red in teakwood from Brazilian fast-growth plantations. Moreover, the study found that both red and yellow pigmentations increased only from 5 to 10 years and then stabilized. This observation is important because according to Gierlinger et al. (2004), teak woods with redder pigment (a*) are more resistant to decay, suggesting that colorimetry can be an indicator of wood quality and durability.
Regarding the saturation (C) and hue angle (h) parameters, the study found that the mean saturation values ranged from 13.6 to 16.9, and the hue angle values ranged from 70.7 to 75.6. The saturation tended to increase with the age of the woods, while the deviation from the 90º angle, indicating the predominance of yellow shade in the material, remained consistent across all ages. The hue angle (h) values ranging from 45 to 90º suggest that the shade of the materials is predominantly yellow for both younger (5 years) and older (20 years) woods.
Thulasidas et al. (2006) reported L* (56.4), a* (6.8), and b* (23.4) values for 35-year-old teak from a plantation in Kerala, India. Derkyi et al. (2009) verified L* (42.9 to 68.3), a* (6.0 to 16.9), and b* (16.8 to 33.3) for teak heartwood from 26 to 50-year-old plantations in different ecological zones of Ghana. Moya and Berrocal (2010) evaluated the color of teak heartwood from trees aged 8 to 15 years, from different site locations in Costa Rica, and found L* (46.8 to 76.5), a* (7.1 to 13.6), and b* (20.1 to 30.1).The present study demonstrated that fast-growth teak plantations, with an age greater than 15 years, can produce wood as dark as plantations with a 35-year age in India. Additionally, wood from fast-growth teak plantations in Brazil was found to have lower levels of yellow and red pigmentation, which results in less saturation compared to previous studies. However, there had been no research measuring the color of the wood from different aged plantations grown in the same location to determine trends in wood color due to age differences, as proposed by Damayanti et al. (2020).
It was found that the colorimetric parameters of teakwood did not vary significantly from 15 to 20 years, which could contribute to a reduction in the final harvest in fast-growth teak plantations. The color of teakwood could also be a significant factor in determining the value and quality of the wood, particularly for darker teakwoods, including older woods, which are in high demand in the market. Overall, these findings provide valuable information about the pigmentation and color changes in teakwood with age, which could be useful for a wide range of applications in the wood industry, from quality control to marketing and product development.
Elemental and Chemical Composition
The elemental and chemical compositions of teakwood varied significantly according to the age of the trees (Table 4). Significant differences were not found only for the contents of hydrogen, sulfur, and total lignin, whose mean values were 6.15%, 0.07, and 32.4%, respectively.
Table 4. Elemental and Chemical Compositions of Teakwood at Different Ages
Caption: Holo. = Holocellulose; Ext. = Total extractives. Values in brackets correspond to standard deviation and lower-case letters in each line indicate significant differences according to the test Tukey (p-value<0.05).
However, the carbon content was found to increase with tree age, while the nitrogen and oxygen contents decreased. These findings are consistent with previous research on the elemental composition of different tree species at varying ages, indicating that carbon content tends to increase as trees grow due to their greater biomass production (Silva et al. 2019) and the higher lignin content of older trees (Table 4). Overall, the results of the elemental composition analysis support those reported in the literature, highlighting the importance of considering tree age when evaluating wood properties. According to Rowell et al. (2013), wood elemental composition, in general, is represented by around 50% carbon, 6% hydrogen, and 44% oxygen, with a small variability among species.
The mean values of total lignin and holocellulose corroborate those found in other research with 4- to 19-year-old teakwood in Brazilian plantations, within the ranges 30.4 to 33.5% and 57.9 to 64.4%, respectively (Floréz 2012; Mascarenhas et al. 2021). The results are also consistent to other studies: Rizanti et al. (2018) assessed logs obtained from the base of teak trees grown in Java, Indonesia, from short (7 to 10 years) and long (40 to 60 years) rotations and they reported a holocellulose content of 68.5% and 67.5%, and a lignin content of 32.2% and 35.5%, respectively; and, Qiu et al. (2019), who evaluated 18-year-old teak from Yunnan, China, which had 26.8 to 27.7% Klason-lignin and 58.3 to 62.2% holocellulose.
The average total extractive values were lower than 10%, which is consistent with the findings of Lukmandaru and Takahashi (2009) (5.3 to 8.0%), Flórez (2012) (8.9%), Rizanti et al. (2018) (3.7 to 8.0%), and Mascarenhas et al. (2021) (3.0%). It should be noted that the percentage of extractives depends on genetic and environmental factors, as well as the type of management and silvicultural techniques employed (Mascarenhas et al. 2021).
There was no consistent trend in the variation of total extractives values with increasing age of the teakwood, and therefore, no direct relationship was observed between heartwood percentage and extractive content. Among the four treatments evaluated, the 15-year-old wood had significantly higher extractive content than the others. This finding contradicts previous studies that proposed a positive correlation between the amount of total extractives in teakwood and its age (Haupt et al. 2003; Thulasidas and Bhat 2007; Lukmandaru and Takahashi 2009). Therefore, only quantitative analysis of the total extractive content is insufficient for accurately inferring the inherent natural durability of teakwood against degradation. Instead, it is necessary to identify and quantify the specific compounds present in the wood in order to establish their relationship with the wood’s durability.
The content of ash varied from 0.9 to 1.5% for five- and 10-year-old woods, respectively. These values were slightly superior to those reported by Flórez (2012) and Mascarenhas et al. (2021), which were 0.6 and 0.7%, respectively. However, these levels of mineral substances are still considered low and are not expected to significantly affect the natural durability of the wood.
GC-MS analysis of the total extractives from teakwood identified sixteen different chemical compounds (Fig. 1 and Table 5). Eight of these compounds were found in both five- and 20-year-old teakwood, while six were only present in five-year-old wood and three, in 20-year-old wood. Although there were no significant differences in extractive percentages between the two age groups (Table 4), their chemical compositions were qualitatively and quantitatively different. The extractives from five-year-old wood were mainly composed of tectoquinone (43.3%), phthalic acid (19.1%), and 1,3-indandione (9.2%), while those from 20-year-old wood were mainly composed of tectoquinone (60.7%), lapachol (13.8%), and phthalic acid (9.7%).
Fig. 1. Gas chromatography–mass spectrometry (GC–MS) chromatogram of the total teakwood extractive at (a) five and (b) twenty years old
The quinone group presented the highest relative percentage, mainly due to the presence of tectoquinone (Lukmandaru and Takahashi 2009; Qiu et al. 2019). Other quinones were also identified in this study: chrysophanol (in 5-year-old wood) and 1,4-hydroxymethyl anthraquinone and lapachol (in 20-year-old wood). Lukmandaru and Takahashi (2009) also correlated the higher content of the components in the quinone group with the older ages of the wood. This group is responsible for wood darkening (Qiu et al. 2019), which may explain why the 20-year-old wood is darker than the 5-year-old wood, since the identified substances are composed of 76.0% and 46.6% quinones, respectively. In addition, the highest concentration of these substances is in the heartwood (Qiu et al. 2019). Table 2 shows 16.3% heartwood for the five-year-old wood, whereas the 20-year-old wood has 60.0% heartwood.
The presence of tectoquinone in teakwood aged less than 10 years is questioned (Moreira et al. 2006). Table 5 and Fig. 1 demonstrate that tectoquinone was the main compound detected through GC-MS for both ages, revealing the capacity of teak to biosynthesize this compound at five years old. However, the presence of lapachol was not detected in the composition of the extracts from the younger wood. It should be noted that naphthoquinones, such as lapachol, are thought to be more effective against fungi than anthraquinones, such as tectoquinone (Sumthong et al. 2008).
Table 5. Chemical Constitution of the Total Extractive in Teakwood at 5 and 20 Years
Caption: a Retention Time; b Identification by comparison of the mass spectra from the reference libraries Adams (2007) and Nist (1990); c Relative percentage of constituents; d Percentage of similarity.
Teak extractives might be a promising source of phytochemicals compounds to be used in the pharmaceutical, cosmetic, or food industries (Chávez-Salgado et al. 2022); in addition to being potential natural wood preservatives (Brocco et al. 2017). In relation to the identified chemical substances with percentages greater than 10%: tectoquinone exhibits strong mosquito larvicidal activity (Cheng et al. 2008), and it is a SARSCoV-2 main protease inhibitor against COVID-19 (Kallingal et al 2022); phthalic acid is an allelochemical agent that can be used as plasticizers and additives to improve various products’ mechanical extensibility and flexibility (Huang et al. 2021); lapachol exhibits antibacterial, antifungal and antitumor activity properties (Marques et al. 2020); and, 1,3-indandione has antibacterial and antifungal properties (Souza 2016), also it is one starting materials for multicomponent reactions for the synthesis of heterocyclic compounds (Asadi and Ziarani 2016).
The extraction of phytochemicals from teakwood extractives of fast-growth plantations presents an opportunity to add value to the wood residues generated during wood industrialization, as well as to the wood from the first thinning and thin logs. This not only provides revenue diversification for teak producers, but it also contributes to sustainable forest management practices by reducing waste and promoting the use of resources. Additionally, the extraction of these compounds from teak extractives can serve as an alternative to synthetic chemicals used in various industries, potentially reducing the environmental impact of these industries.
Deterioration of Teak Wood in Accelerated Decay Test
The mass loss in the reference wood (J. copaia) after 16 weeks of testing was 56.3% in a decay test by the fungus T. versicolor. These values were higher than the minimum value of 50% established by the D-2017 standard (ASTM, 2005). Therefore, the result attests that the general testing conditions were favorable for the growth of the fungus and that culture was in full vegetative vigor, confirming the validity of the data shown in Fig. 2.
Fig. 2. Mass loss in the pith-bark direction of teakwood aged 5 and 20 years after 16 weeks of accelerated decay test using Trametes versicolor. Bars correspond to standard deviation, lower-case letters indicate significant differences for 5 years old, and upper-case letters indicate significant differences for 20 years old, along the radius, according to the Tukey test (p-value<0.05).
The 5-year-old samples were classified as resistant regardless of the radial profile, with an average mass loss of 21.9% (Fig. 2). Although there was a significant loss of mass, teakwood from the first thinning already exhibited some resistance to biodeterioration. Based on extractive analysis results (Table 5), five-year-old teak already biosynthesizes tectoquinone, a compound that contributes to its natural durability. It is interesting to note that both the heartwood and sapwood of teakwood showed similar natural durability. This result differs from that of Oliveira et al (2019), who evaluated the biological resistance of the sapwood and heartwood of six-year-old teakwood and classified the sapwood as “slightly resistant” (40.8%) and the heartwood as “resistant” (18.9%). However, the result obtained in this study can be explained by the presence of tectoquinone in the sapwood (Niamké et al. 2011; Qiu et al. 2019).
This suggests that teakwood from early thinning could potentially be used for certain applications where high durability is not a critical requirement, such as indoor furniture, small decorative items, or interior finishings. It is important to note that the use of teakwood from thinning can contribute to the promotion of fast-growing plantations.
The 20-year-old teakwood showed significant variation in mass loss depending on the region, with positions closer to the pith and sapwood being classified as resistant, and the remaining positions corresponding to heartwood were highly resistant with mass losses below 10% (Fig. 2). The lower durability in the pith region might be explained by the loss of extractive toxicity over time, thus making the wood more susceptible to biodeterioration (Motta et al. 2013). Another possible explanation could be that the wood cell wall is less thick in the early years, which can facilitate the development of xylophagous fungi. The extractive content of teakwood that remains concentrated in the heartwood region provides the wood with greater durability than the sapwood (Brocco et al. 2017).
These results are similar to those obtained by Brochini (2019), who assessed the biodeterioration of 20-year-old teakwood subjected to the action of T. versicolor for 60 days and found a mean mass loss of 5.19% for heartwood and 8.55% for sapwood. Brocco et al. (2017) also observed a low mass loss (8.88%) for a 20-year-old teakwood for the same fungus over 12 weeks. It is worth noting that of the components identified in the extractives of 20-year-old teakwood, approximately 75% were composed of tectoquinone and lapachol (Table 5), which justifies its greater resistance to deterioration compared to the younger wood.
For outdoor applications, such as decking or outdoor furniture, it is still advisable to use the heartwood from final harvesting fast-growth plantations, which has higher natural durability than wood from thinning. While the 20-year-old teakwood showed high resistance to biodeterioration, it is important to note that the study only assessed the resistance of the wood to a specific fungus and under specific conditions. Other factors such as exposure to weather, insects, and fungi in different environments may impact the durability of the wood. Therefore, it is important to consider the intended use and environmental conditions when selecting the appropriate wood for a project.
CONCLUSIONS
- When the age changes from five to twenty years, the diameter of the base of the trees doubles while the diameter and percentage of the heartwood triples. However, when comparing the ages of 15 and 20 years, these parameters have an increase of less than 10%.
- As age advances, teakwood becomes darker and more saturated, and the predominance of yellow color is lower than red.
- The carbon content increases as the tree ages, followed by lower contents of nitrogen and oxygen. The percentage of total lignin is 32.4%, without significant variations as age advances.
- There is no clear trend of increasing total extractive content as age advances. However, the chemical composition and proportion of compounds change, with tectoquinone becoming predominant 43.3% in 5-year-old wood and 60.7% in 20-year-old wood, respectively.
- The five-year-old wood is classified as resistant to the action of wood decay fungi (Trametes versicolor), regardless of base position. The 20-year-old wood, in turn, is very resistant in its heartwood region and resistant in its regions of sapwood and heartwood close to the pith.
- The demand for teakwood from fast-growth plantations is expected to increase, given the colorimetric parameters, chemical properties, and biological resistance that the species already exhibits in younger wood. The 5- and 10-year-old teak is best suited for manufacturing small indoor objects. Teak heartwood from older age classes (15 to 20 years) is recommended for producing higher value-added products, especially those intended for outdoor use. Additionally, the extraction of phytochemicals is an opportunity to add value to the teak production chain, through the use of wood waste as well as thin logs resulting from forest thinning.
ACKNOWLEDGMENTS
The authors acknowledge the Coordination for the Improvement of Higher Education Personnel (CAPES) for the first author’s master’s scholarship and for the payment of the publication fee.
REFERENCES CITED
Adams, R. P. (2007). Identification of Essential Oil Components by gas Chromatography/ Mass Spectrometry (4th Ed.), Allured Publishing Corporation, Carol Stream, IL, USA.
Amusant, N., Beauchene, J., Fournier, M., Janin, G., and Thevenon, M. F. (2004). “Decay resistance in Dicorynia guianensis Amsh.: Analysis of inter-tree and intra-tree variability and relations with wood colour,” Annals of Forest Science 61(4), 373-380. DOI: 10.1051/forest:2004030
Asadi, S., and Ziarani, G. M. (2016). “The molecular diversity scope of 1,3-indandione in organic synthesis,” Molecular Diversity 20, 111-52. DOI: 10.1007/s11030-015-9589-z.
ASTM D1102-84 (2013). “Ash in wood,” ASTM Intl., West Conshohocken, PA, USA.
ASTM D1105-96 (2013). “Method for preparation of extractive-free wood,” ASTM International, West Conshohocken, PA, USA.
ASTM D-2017 (2005). “Accelerated laboratory test of natural decay resistance of woods,” ASTM International, West Conshohocken, PA, USA.
Brocco, V. F., Paes, J. B., da Costa, L. G., Brazolin, S., and Arantes, M. D. C. (2017). “Potential of teak heartwood extracts as a natural wood preservative,” Journal of Cleaner Production 142, 2093-2099. DOI: 10.1016/j.jclepro.2016.11.074
Brochini, G.G. (2019). Biodeterioration of Teak Wood (Tectona grandis L.) Subjected to Attack by Lignocellulolytic Fungi, Master’s Thesis, Federal University of Paraná, Curitiba, Paraná, Brazil. (in Portuguese)
Camargos, J. A. A., and Gonçalez, J. C. (2001). “Applied colorimetry as instrument in the elaboration of a timber color chart (in Portuguese),” Brasil Florestal 71, 30-41.
Camino, R., and Morales, J. P. (2013). Teak Plantations in Latin America: Myths and Realities (Technical Series No. 397), Tropical Agricultural Research and Higher Education Center – CATIE, Turrialba, Costa Rica. (in Spanish)
Chávez-Salgado, L. P., Vandenbossche, V., and Vilarem, G. (2022). “Tectona grandis Linn. f. secondary metabolites and their bioactive potential: A review,” iForest: Biogeosciences and Forestry 15(2), 112-120. DOI: 10.3832/ifor3714-015
Cheng, S. S., Huang, C. G., Chen, W. J., Kuo, Y. H., and Chang, S. T. (2008). “Larvicidal activity of tectoquinone isolated from red heartwood-type Cryptomeria japonica against two mosquito species,” Bioresource Technology 99, 3617–3622. DOI: 10.1016/j.biortech.2007.07.038
Damayanti, R., Ozarska, B., Ilic, J., Pari, G., Dwianto, W., Indrawan, D. A., and Krisdianto, K. (2020). “Variation of heartwood proportion and wood colour from fast grown 5-year-old teak,” Wood Research Journal 11, 1-11. DOI: 10.51850/wrj.2020.11.1.1-11
Derkyi, N. S. A., Bailleres, H., Chaix, G., Thevenon, M. F., Oteng-Amoako, A. A., and Adu-Bredu, S. (2009). “Colour variation in teak (Tectona grandis) wood from plantations across the ecological zones of Ghana,” Ghana J. Forestry 25, 40-48.
Flórez, J. (2012). Technologic Characteristics of Young Wood of Tectona grandis Lf, Master’s Thesis, Federal University of Lavras, Minas Gerais, Brazil. (in Portuguese)
Gaitan-Alvarez, J., Moya, R., and Berrocal, A. (2019). “The use of X-ray densitometry to evaluate the wood density profile of Tectona grandis trees growing in fast-growth plantations,” Dendrochronologia 55, 71-79. DOI: 10.1016/j.dendro.2019.04.004
Gierlinger, N., Jacques, D., Grabner, M., Wimmer, R., Schwanninger, M., Rozenberg, P., and Pâques, L. E. (2004). “Colour of larch heartwood and relationships to extractives and brown-rot decay resistance,” Trees 18, 102-108. DOI: 10.1007/s00468-003-0290-y
Goldschmid, O. (1971). “Ultraviolet spectra,” in Lignins: Occurrence, Formation, Structure and Reactions, K. V. Sarkanen, C. H. Ludwig (eds.), Wiley Interscience, New York, NY, USA, pp. 241-266.
Gomide, J. L., and Demuner, B. J. (1986). “Determination of lignin content in woody material: modified Klason method,” O Papel 47(8), 36-38.
Haupt, M., Leithoff, H., Meier, D., Puls, J., Richter, H.G., and Faix, O. (2003). “Heartwood extractives and natural durability of plantation-grown teakwood (Tectona grandis L.f.) – a case study,” Holz als Roh- und Werkstoff 61, 473-474.
Huang, L., Zhu, X., Zhou, S., Cheng, Z., Shi, K., Zhang, C., and Shao, H. (2021). “Phthalic acid esters: Natural sources and biological activities,” Toxins 13(7), 495. DOI: 10.3390/toxins13070495
IBÁ (2019). Report 2019, Brazilian Tree Industry, Brasília, DF, Brazil.
ITTO (2022). Tropical Timber Market Report (Report Volume 26, No 19), International Tropical Timber Organization, Yokohama, Japan.
Kallingal, A., Thachan Kundil, V., Ayyolath, A., Karlapudi, A. P., Muringayil Joseph, T., and Variyar, E, J. (2022). “Molecular modeling study of tectoquinone and acteoside from Tectona grandis Linn: A new SARS-CoV-2 main protease inhibitor against COVID-19.” Journal of Biomolecular Structure and Dynamics 40, 1764-1775. DOI: 10.1080/07391102.2020.1832580
Kollert, W., and Kleine, M. (2017). The Global Teak Study: Analysis, Evaluation and Future Potential of Teak Resources (IUFRO World Series Volume 36), International Union of Forest Research Organizations (IUFRO), Vienna, Austria.
Leite, H. G., Oliveira, N., Monte, M. A., Fardin, L., de Alcantara, A. M., Binoti, M. D. S., and Castro, R. V. O. (2011). “Taper models of heartwood of Tectona grandis Lf.,” Scientia Forestalis 39(89), 53-59.
Lukmandaru, G., and Takahashi, K. (2009). “Radial distribution of quinones in plantation teak (Tectona grandis Lf),” Annals of Forest Science, 66(6), 1-9. DOI: 10.1051/forest/2009051
Marques, L. B., Ottoni, F. M., Pinto, M. C. X., Ribeiro, J. M., de Sousa, F. S., Weinlich, R., et al. (2020) “Lapachol acetylglycosylation enhances its cytotoxic and pro-apoptotic activities in Hl60 cells,” Toxicology in Vitro 65, 1-13. DOI: 10.1016/j.tiv.2020.104772
Mascarenhas, A. R. P., Sccoti, M. S. V., Melo, R. R. D., Corrêa, F. L. D. O., Souza, E. F. M. D., and Pimenta, A. S. (2021). “Quality assessment of teak (Tectona grandis) wood from trees grown in a multi-stratified agroforestry system established in an Amazon rainforest area,” Holzforschung, 75(5), 409-418. DOI: 10.1515/hf-2020-0082
Mehats, J., Castets, L., Grau, E., and Grelier, S. (2021). “Homogenization of maritime pine wood color by alkaline hydrogen peroxide treatment,” Coatings 11(7), 839-856. DOI: 10.3390/coatings11070839.
Moreira, R. Y., Arruda, M. S., Arruda, A. C., Santos, L. S., Müller, A. H., Guilhon, G. M. S. P, Santos, A. S., and Terezo, E. (2006). “Anthraquinones and naphthoquinones from the stem of a reforested specimen of Tectona grandis (Verbenaceae),” Revista Brasileira de Farmacognosia 16, 392-396. (in Portuguese)
Motta, J. P., Oliveira, J. T. S., Paes, J. B., Alves, R. C., and Dambroz, G. B. V. (2013) “Natural resistance of Tectona grandis wood in laboratory tests (in Portuguese),” Ciência Rural 43(8), 1393-1398. DOI: 10.1590/S0103 84782013005000097
Moya, R., and Berrocal, A. (2010). “Wood colour variation in sapwood and heartwood of young trees of Tectona grandis and its relationship with plantation characteristics, site, and decay resistance,” Annals of Forest Science 67, 109-09. DOI:10.1051/forest/2009088
Moya, R., Bond, B., and Quesada, H. (2014). “A review of heartwood properties of Tectona grandis trees from fast-growth plantations,” Wood Science and Technology 48(2), 411-433. DOI: 10.1007/s00226-014-0618-3.
Niamké, F. B., Amusant, N., Charpentier, J. P., Chaix, G., Baissac, Y., Boutahar, N., Adima, A. A., Kati-Coulibaly, S., and Jay-Allemand, C. (2011). “Relationships between biochemical attributes (non-structural carbohydrates and phenolics) and natural durability against fungi in dry teak wood (Tectona grandis L. f.),” Annals of Forest Science 68, 201-211. DOI: 10.1007/s13595-011-0021-2
NIST 05 Spectral Library (1990). “NIST Mass Spectrometry,” National Institute of Standards and Technology, Washington, DC, USA.
Oliveira, W. C., Pereira, B. L. C., Goes, L. S. D., Quintilhan, M. T., Oliveira, A. C., and Mora, R. (2019). “Deterioration of Teak wood in accelerated decay test,” Floresta e Ambiente 26 (2), 1-9. DOI: 10.1590/2179-8087.036017.
Pereira, B. L. C., Oliveira, A. C., Carvalho, A. M. M. L., Carneiro, A. C. O., Vital, B. R., and Santos, L. C. (2013). “Correlations among the heart/sapwood ratio of Eucalyptus wood, yield and charcoal properties (in Portuguese),” Scientia Forestalis 41(98), 217-225.
Qiu, H., Liu, R., and Long, L. (2019). “Analysis of chemical composition of extractives by acetone and the chromatic aberration of teak (Tectona grandis LF) from China,” Molecules 24(10), 1989-1998. DOI: 10.3390/molecules24101989
Ribeiro, E. S. (2017). Technological Properties of Twenty Species of Tropical Wood Commercialized by the State of Mato Grosso, Ph.D. Dissertation. Federal University of Brasília, Brasília, Distrito Federal, Brazil. (in Portuguese)
Rizanti, D. E., Darmawan, W., George, B., Merlin, A., Dumarcay, S., Chapuis, H., Gerardin, C., Gelhaye, E., Raharivelomanana, P., Sari, R. K., et al. (2018). “Comparison of teak wood properties according to forest management: Short versus long rotation,” Annals of Forest Science 75(39), 1-12. DOI: 10.1007/s13595-018-0716-8
Rowell, R. M., Pettersen, R., and Tshabalala, M. A. (2013). “Cell wall chemistry,” in: Handbook of Wood Chemistry and Wood Composites, M. R. Rowell (ed.), CRC Press, Boca Raton, FL, USA, pp. 33-72.
Sanquetta, M. N. I., Sanquetta, C. R., Corte, A. P. D., Mognon, F., and Behling, A. (2014). “Diametric increment and percentages of heartwood and bark in Tectona grandis L F stands in Southeast Pará,” Enciclopedia Biosfera 10(18), 1946-1954. (in Portuguese)
Schneider, C.A., Rasband, W.S., Eliceiri, K.W. (2012). “NIH Image to ImageJ: 25 years of image analysis,” Nature Methods 9, 671-675.
Silva, E. J. D., Cremonez, V. G., and Nisgoski, S. (2019). “Color change and thermogravimetric analysis of thermally treated Eucalyptus grandis wood,” Revista Ciência da Madeira 10(1), 39-47. DOI: 10.12953/2177-6830/rcm.v10n1p39-47.
Siqueira, R. G., Evangelista, W. V., Ferreira, J. L., and Garlet, J. (2020). “Natural resistance of Teak wood to termite attack Cornitermes bequaerti Emerson, 1952,” in: Forest Engineering in Focus: Challenges, Limits and Potentialities, R. J. Oliveira (Org.), Editora Científica, Guarujá, SP, Brazil, pp. 759-778. (in Portuguese)
Souza, A. M. (2016). Synthesis of 2-arylidene indan-1, 3-diones and Evaluation of Antiviral, Cytotoxic and Leishmanicidal Activities, Master’s Thesis, Federal University of Viçosa, Viçosa, Minas Gerais, Brazil. (in Portuguese)
Sumthong, P., Romero-Gonzales, R., and Verpoorte, R. (2008). “Identification of anti-wood rot compounds in teak (Tectona grandis L.f.) sawdust extract,” Journal of Wood Chemistry and Technology 28, 247–260.
Takizawa, F. H. (2022). “Use of wood,” in: Teak: From Planting to Harvesting, F. H. Takizawa, R. A. Medeiros, H. G. Leite, A. Borém (eds.), Editora UFV, Viçosa, MG, Brazil, pp. 318-344. (in Portuguese)
Thulasidas, P. K., and Bhat, K. M. (2007). “Chemical extractive compounds determining the brown-rot decay resistance of teak wood,”, Holz Roh-Werkstoff 65, 121-124.
Thulasidas, P. K., Bhat, K. M., and Okuyama, T. (2006). “Heartwood colour variation in home garden teak (Tectona grandis) from wet and dry localities of Kerala, India,” Journal of Tropical Forest Science 18(1), 51-54.
Ugalde Arias, L. A. (2013). TEAK: New Trends in Silviculture, Commercialization and Wood Utilization, International Forestry and Agroforestry, San Jose, Costa Rica.
Article submitted: March 16, 2023; Peer review completed: April 1, 2023; Revised version received and accepted: April 18, 2023: Published: April 26, 2023.
DOI: 10.15376/biores.18.2.4116-4131
