Abstract
The capacities of white rot fungal treatments to degrade lignocellulose and further to improve the rumen degradability were examined in two kinds of corn stover (CAU3138 and ZN01). A total of seven fungi were evaluated according to their growth rates and selective values (SV) on both stovers under 28 °C for 28 days. Then, three selected fungi were evaluated by their in vitro gas production (IVGP) as a measure of rumen fermentation capacity. The results showed that Irpex lacteus, Pleurotus ostreatus, and Pleurotus cystidiosus had high speeds of colonization and high SV on both stovers. The IVGP of the ZN01 treated with any of the three fungi was lower than that of the raw stover, and the IVGP of the CAU3138 treated with P. ostreatus and P. cystidiosus had no significant change (P > 0.05). However, the IVGP of the CAU3138 treated with I. lacteus was significantly increased (P < 0.05) and further increased as the treatment time was prolonged. This study indicated that SV was not always a good predictor for the performance of the fungus. Here, I. lacteus performed best, regarding fungal growth and IVGP of the CAU3138.
Download PDF
Full Article
Effect of White-rot Fungal Treatments on the in Vitro Rumen Degradability of Two Kinds of Corn Stover
Sasa Zuo, Dongze Niu, Di Jiang, Pengjiao Tian, Rongrong Li, Wei Wu, and Chuncheng Xu *
The capacities of white rot fungal treatments to degrade lignocellulose and further to improve the rumen degradability were examined in two kinds of corn stover (CAU3138 and ZN01). A total of seven fungi were evaluated according to their growth rates and selective values (SV) on both stovers under 28 °C for 28 days. Then, three selected fungi were evaluated by their in vitro gas production (IVGP) as a measure of rumen fermentation capacity. The results showed that Irpex lacteus, Pleurotus ostreatus, and Pleurotus cystidiosus had high speeds of colonization and high SV on both stovers. The IVGP of the ZN01 treated with any of the three fungi was lower than that of the raw stover, and the IVGP of the CAU3138 treated with P. ostreatus and P. cystidiosus had no significant change (P > 0.05). However, the IVGP of the CAU3138 treated with I. lacteus was significantly increased (P < 0.05) and further increased as the treatment time was prolonged. This study indicated that SV was not always a good predictor for the performance of the fungus. Here, I. lacteus performed best, regarding fungal growth and IVGP of the CAU3138. Keywords: White-rot fungi; Corn stover; Selective value; In vitro gas production; Acid detergent lignin degradation; Irpex lacteus
Contact information: College of Engineering, China Agricultural University, Beijing 100083, China;
* Corresponding author: xucc@cau.edu.cn
INTRODUCTION
Corn stover is an attractive renewable source of roughage for ruminant animals. In China, approximately 300 million tons of corn stover are produced annually (Chen et al. 2017). However, corn stover has a low degradability. Consequently, most of the corn stover has been abandoned in the field or burnt directly, heavily polluting the environment. Within corn stover cell walls, acid detergent lignin (ADL), a large group of recalcitrant aromatic polymers bonded with hemicellulose by ether and ester linkages, forms a matrix that tightly surrounds the cellulose. This complex lignocellulosic structure inhibits the fermentation process by rumen microbes and hydrolyzation by cellulose (Van Kuijk et al. 2015b). Therefore, it is necessary to disrupt the covalent bonds between ADL and hemicellulose via treatment to release the cellulose from this matrix and enhance the feed utilization rate of corn stover. Moreover, development and utilization of corn stover in animal husbandry have become important for sustainable agriculture development.
Biological treatment, especially white-rot fungal treatment, has been regarded as an environmentally friendly and potentially economically viable alternative to physical and chemical treatments to improve the in vitro rumen degradability of the lignocellulose biomass for the ruminants (Tuyen et al. 2013; Van Kuijk et al. 2015a). However, white-rot fungi have different abilities and methods of degrading lignocellulose, and some take a relatively long time to germinate their mycelium. Moreover, white-rot fungi consume a major portion of cellulose and hemicellulose as part of the fungus’ growth. For example, although Phanerocheate chrysosporium is a well-known white-rot fungus that effectively degrades ADL, it is a non-selective fungus that consumes large quantities of cellulose and hemicellulose during its degradation of lignocellulosic biomass (Syed and Yadav 2012). Therefore, it is necessary to select fungi with high growth speeds that can inhibit mould and bacterial contamination (Tirado-González et al. 2016). In doing so, one should use a selective value (SV; ratio of ADL degradation to cellulose degradation) to screen fungi that can selectively degrade the corn stover (Tuyen et al. 2012; Saha et al. 2016). In contrast, some researchers have reported that moderate cellulose degradation can increase glucose recoveries because increased glucose recovery also contributes to the degradation of ADL (Salvachúa et al. 2011; Niu et al. 2018). That is to say, treatments with the highest ADL loss, rather than selective ADL degradation, have the greatest potential for increasing the nutritional value of the feed (Rahman et al. 2011). Before now there have been no certain conclusions about whether or not SV is a crucial parameter in evaluating the nutritional value of corn stover with white-rot fungal treatment. Thus, the role of SV in improving the rumen degradability of corn stover with fungal treatment should be further examined. To study the improvement in rumen degradability by white rot fungal treatment. The in vitro gas production (IVGP) technique was used. Menke et al. (1979) reported a high correlation between gas production in vitro and in vivo apparent digestibility. The IVGP technique has been widely used for evaluation of nutritive value of feed.
In the present study, seven species of fungi were used on corn stovers CAU3138 and ZN01. The CAU3138 corn stover is from the grain and forage corn variety that is tall and strong with higher ADL content. The ZN01 corn stover is from the waxy corn variety and is short and weak with lower ADL content. The objectives of the present study were: (1) to examine the differences between seven fungal species on selective ADL degradation in the corn stovers CAU3138 and ZN01; (2) to employ the IVGP technique to compare the ability of the selected fungi on the improvement of the nutritional value of CAU3138 and ZN01; and (3) based on the IVGP results, to further study the effects of two different particle sizes (1 mm and 2 cm) with a prolonged treatment time of 42 days on CAU3138 and the optimization of these parameters to obtain a high in vitro rumen degradability.
EXPERIMENTAL
“Experiment 1” was designed to preliminarily screen for the fungal strains with high growth rates and SV values. Then, the in vitro rumen degradabilities of the two kinds of corn stovers treated with the preliminarily screened fungi, as measured by the IVGP technique, were compared. Based on the results of IVGP in Experiment 1, Experiment 2 was designed to optimize the screened fungus treatments of corn stover CAU3138 with an extended treatment time and enlarged particle sizes.
Materials
Substrate preparation
Corn stover CAU3138 (approximately 110 d from sowing to harvesting) is from the grain and forage corn cultivar; corn stover ZN01 (approximately 90 d from sowing to harvesting) is from the waxy corn variety. Both stovers were collected from local farmlands at Baoding City, Hebei Province, China. The whole aboveground plant was initially air-dried at room temperature for one month. Both feedstuffs were chopped into average particle sizes of 2 cm in length. A portion was milled in RT-34 hammer mill (Rong Tsong Precision Technology Co., Taiwan). and passed through a 1-mm screen. Both processed corn stovers were then sealed in plastic bags and stored at room temperature for further use. The chemical compositions of CAU3138 and ZN01 are shown in Table 1.
White-rot fungal strains and spawn preparation
The three white-rot fungal strains used in the present study (Phanerochaete chrysosporium (CGMCC-5.776), Irpex lacteus (CGMCC-5.809), and Pleurotus ostreatus (CGMCC-5.374)) were purchased from the China General Microbiological Culture Collection Center (CGMCC) in Beijing, China.
Table 1. Chemical Compositions of the Raw and Autoclaved CAU3818 and ZN01
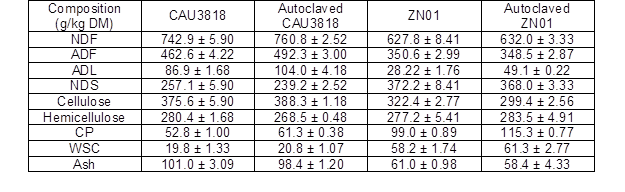
Note: NDF- neutral detergent fiber; ADF- acid detergent fiber; ADL- acid detergent lignin; NDS- acid detergent soluble; CP- crude protein; WSC- Water soluble carbohydrate; data are mean ± standard error
In addition, the following four Basidiomycete strains were obtained from the Shangzhuang Mushroom Breeding Center (Beijing, China), isolated in the authors’ laboratory, and identified via rDNA internal transcribed spacer (ITS) sequence analysis. These strains were: Pleurotus cystidiosus (accession number: KY828220), Gymnopus perforans (KY848622), Flammulina velutipes (KY828221), and Pleurotus ferulaginis (MF067417). All these basidiomycete strains were maintained in the laboratory on Potato Dextrose Agar (PDA) slants at 4 °C. Initial cultures of these fungi were developed on PDA plates at 28 °C until mycelium covered the entire agar plates. A total of 20 pieces of colonized agar cultures (approximately 1 cm2) of I. lacteus were separately added to sterilized wheat grains and incubated at 28 °C until all grains were colonized by mycelium. The spawns were kept at 4 °C until further use.
Methods
Fungi treatments of 1-mm corn stover CAU3138 and ZN01 for 28 days (experiment 1)
The sterilization and fungal inoculation processes were performed on 1-mm substrates CAU3138 and ZN01 according to Zuo et al. (2018). The inoculated substrates and the control (autoclaved but un-inoculated substrates) were incubated at 28 °C for 7, 14, 21, and 28 days in an air-conditioned chamber with 85% relative humidity. Triplicates of the 1-mm substrates treated with individual fungi plus the petri dishes were taken after the elapsed time, weighed, and oven-dried at 65 °C until constant weight for dry matter (DM) loss determination.
In vitro gas production technique
In vitro gas production was performed according to the procedure described by Menke et al. (1979). In summary, rumen fluid was collected in the morning (before feeding) from three 3-year old rumen-fistulated Angus bullocks fed a corn silage and wheat straw-based diet at the China Agricultural University Beef Cattle Breeding Center (Beijing, China). The samples were then immediately transferred to the laboratory in a thermos bottle. The fresh rumen fluid was mixed with a buffer solution in a 1:2 (v/v) ratio under a continuous flux of CO2. Approximately 220 mg of the oven-dried corn stover substrates (already treated with each fungus) was weighed into each of the three 100-mL calibrated glass syringes. The glass syringes were pre-warmed to 39 °C, then 30 mL of mixed culture medium were pipetted with an automatic pump into each glass syringe, and then they were incubated in a water bath at 39 °C. A control was included that contained corn stover that had not been treated with any of the fungi. A blank glass syringe only contained 30 mL of mixed culture medium. All the cultures had three replicates. The gas production was manually recorded at 0, 2, 4, 6, 8, 10, 12, 16, 20, 24, 30, 36, 42, 48, 60, 72, 84, and 96 h, calculated by subtracting the volumes of gas in the blank calibrated syringes, and expressed on an OM basis.
Optimization of I. lacteus treatment of 1-mm and 2-cm CAU3138 Stover for 42 days (experiment 2)
The methods used in experiment 2 were the same as those used in experiment 1. The first part of experiment 2 investigated the effects of an extended treatment time (42 days) and the use of a smaller CAU3138 particle size (1 mm) on IVGP by I. lacteus. The second part of experiment 2 was followed as: the moisture content of the 2-cm CAU3138 particles was also adjusted to 70% by adding 642 mL distilled water to 300 g air-dried corn stover (DM 942 g/kg). After thorough mixing, 100 g substrates were weighed in polypropylene bags and sterilization and fungal inoculation processes were performed according to Van Kuijk et al. (2015b). The substrate was aseptically inoculated with 4 g of previously prepared wheat spawn of I. lacteus. The bags of inoculated CAU3138 along with the control were incubated in an air-conditioned chamber at 28 °C for 7, 14, 28, and 42 days. The samples of 2-cm CAU3138 (with bags) were taken at the specified times, weighed, and oven-dried at 65 °C until constant weight; then, the DM of the 2-cm CAU3138 particles was ground to 1 mm and preserved for chemical composition determination and IVGP measurement. All of the treatments were performed in triplicate.
Chemical analysis
The neutral detergent fibre (NDF) content was analyzed by the modified method of Van Soest et al. (1991) with the addition of a heat stable amylase but without the addition of sodium sulphite. The acid detergent fiber (ADF) content and ADL content were analyzed via the method of Van Soest (1973). Neutral detergent solute (NDS) content was calculated by subtracting the NDF from 1000. The hemicellulose (HC) content was calculated as the difference between the NDF and ADF, and the cellulose (Cell) content was calculated as the difference between the ADF and ADL. The water soluble carbohydrate (WSC) content was analysed using the anthrone-sulphuric acid colorimetry method (Ning et al. 2017). The crude protein (CP) content of the substrates was tested according to the Kjeldahl method. Ash content was determined by combustion for 3 h at 550 °C in a muffle furnace according to Van Kuijk et al. (2015b).
Statistical Analysis
The results of SV, chemical loss analysis, and IVGP data of the fungal treatment of each substrate were subject to a generalized linear model (GLM) analysis in SAS 9.2 (SAS Institute Inc., Cary, NC, USA) software. The following model was used,
![]() (1)
(1)
where Yij is the observation j in treatment i, µ is the overall mean, αi is the fixed effect of treatment i, and ωij is the random error.
The effect of incubation time and particle size on the detergent fiber composition and IVGP of CAU3138 was tested using the generalized linear model (GLM) analysis in SAS 9.3 software (SAS Institute Inc., Cary, NC, USA). The following model was used,
![]() (2)
(2)
where Yij is the observation at incubation time i, µ is the overall mean, αi is the fixed effect of incubation time i, βj is the fixed effect of particle size j, and ωij is the random error.
RESULTS AND DISCUSSION
Seven Fungi Treatment of CAU3138 and ZN01
All fungi used in the present study were able to grow on CAU3138 and ZN01. In the early stage of incubation, rapid growth of P. ostreatus, I. lacteus, P. chrysosporium, and P. ferulaginis on the corn stover CAU3138 was observed. After two weeks, mycelium of the above four fungi almost covered all of the CAU3138 substrates and P. cystidiosus began to grow quickly at this time. For F. velutipes- and G. perforans-treated CAU3138, very limited amounts of mycelium were visually observed. Different from CAU3138, all of the fungi were able to grow rapidly on the ZN01 and almost covered the entire petri dishes during the initial two weeks.
Table 2. Component Loss (%) and Selectivity Values (ADL Degradation/ Cellulose Degradation) of 1-mm Corn Stover CAU3138 and ZN01 After 28 Days Incubation with Seven White-rot Fungi
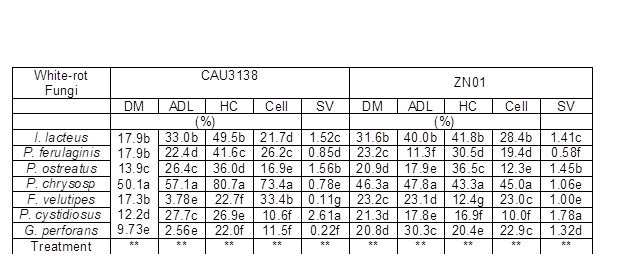
Note: P. chrysosp-Phanerochaete chrysosporium; Means of each fungus treatment within a column that are followed by the same letter are not significantly different from each other at P < 0.05; **- P < 0.001; DM- dry matter; ADL- acid detergent lignin; HC- hemicelluloses; Cell- cellulose It was obvious that the ZN01 had higher nutritional value than the CAU3138 (Table 1). The cellulose and ADL content of the ZN01 was lower than that of CAU3138; moreover, the NDS content of ZN01 was higher than that of CAU3138 and its WSC content was almost three-fold that of the CAU3138. All fungi showed a net loss in DM, cellulose, hemicellulose, and ADL when on the 1-mm CAU3138 and ZN01 substrates (Table 2). For both stovers, the incubations with P. ostreatus, P. cystidiosus, and G. perforans showed the lowest losses of DM, whilst the incubations with P. chrysosporium showed the highest losses of DM (P < 0.01). The highest loss of ADL in the CAU3138 and ZN01 was due to P. chrysosporium treatment, followed by I. lacteus treatment (P < 0.01). The P. chrysosporium and I. lacteus incubations also resulted in higher losses (P < 0.01) of hemicellulose in both stovers than other fungi, yet losses of cellulose due to I. lacteus treatment were much lower than P. chrysosporium treatment. Pleurotus ostreatus consumed a small percentage of cellulose but a relatively large proportion of hemicellulose of both stover. However, incubation with P. cystidiosus led to the lowest loss of cellulose for both stovers (P < 0.01). In this study, after 28 days of treatment, the SV of CAU3138 treated by all seven strains varied from 0.11 to 2.61, while the SV of ZN01 varied from 0.58 to 1.78 (Table 2). The CAU3138 and ZN01 samples treated with P. ostreatus, P. cystidiosus, and I. lacteus were completely covered by their mycelium after 28 days and showed higher SV (following the order: P. cystidiosus > P. ostreatus > I. lacteus) after 28 days of treatment. Therefore, P. ostreatus, P. cystidiosus, and I. lacteus were selected for the further study.
In vitro Gas Production
Raw CAU3138 corn stover showed an IVGP value of 177.9 mL/g OM, while the IVGP of the 1-mm CAU3138 control was 199.3 mL/g OM, making this a significant increase compared with the raw material (P < 0.01). The gas production kinetic parameters estimated using Eq. 1 revealed that the rate of gas production was significantly (P < 0.01) improved after 28 days of treatment of the 1-mm CAU3138 with I. lacteus and P. cystidiosus. However, only the 1-mm CAU3138 treated with I. lacteus for 28 days had significantly (P < 0.01) greater asymptotic gas production, which resulted in an increased (P < 0.05) IVGP of 66.8 mL/g OM (33.5% increase). There was no increase in total IVGP for the 1-mm CAU3138 treated by P. ostreatus or P. cystidiosus (Fig. 1a). Raw ZN01 corn stover had an IVGP value of 278.3 mL/g OM. The IVGP of the ZN01 control was 273.0 mL/g OM; thus, autoclave sterilization had no significant effect on the gas production of ZN01 (P > 0.05). Moreover, IVGP decreased (P < 0.01) after the three fungi treatments with lower asymptotic and rate of gas production than the control of ZN01 (Fig. 1b).
When the treatment time was extended to 42 days for the 1-mm CAU3138, IVGP for the sample treated with I. lacteus increased significantly (P < 0.01) (Fig. 1a and 1c). For the CAU3138 stover with a 2-cm particle size, IVGP of the sample treated with I. lacteus did not increase compared with the 1-mm stover treated for the same periods (Fig. 1a and 1d). Interestingly, the smaller particle size was beneficial for the gas production of the I. lacteus treatment.
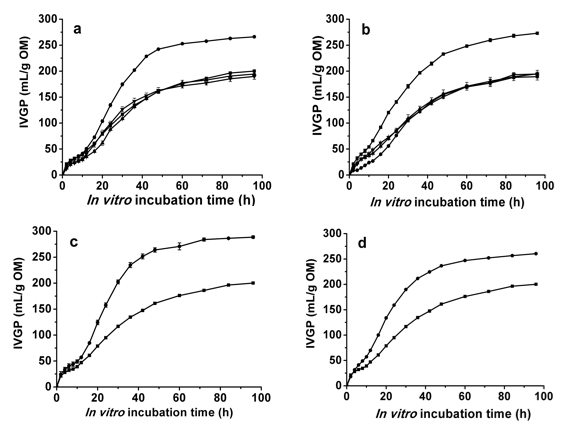
Fig. 1. Results of IVGP measurements after the selected fungi treatment of corn stover compared with the control (autoclaved but un-inoculated substrates); (a) Three fungi treatment of 1-mm CAU3138 for 28 days, (b) Three fungi treatment of 1-mm ZN01 for 28 days, (c) Irpex lacteus treatment of 1-mm CAU3138 for 42 days, (d) Irpex lacteus treatment of 2-cm CAU3138 for 42 days; (■) Control, (●) Irpex lacteus, (▲) Pleurotus ostreatus, (▼) Pleurotus cystidiosus; error bars indicate standard deviations, n = 3
Chemical Analysis of I. lacteus Treatment of the 1-mm and 2-cm CAU3138 for 42 Days
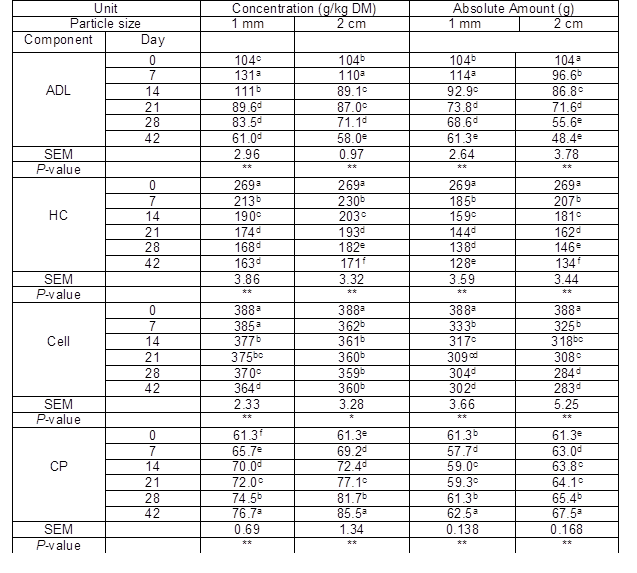
As no improvement in the IVGP of the ZN01 stover treated by the three selected fungi (relative to the control) was observed, no further studies of ZN01 were conducted. Table 3 shows the changes of detergent fiber and CP of CAU3138 during the 42 days of incubation with I. lacteus. This fungus caused a decrease (P < 0.05) in ADL concentration (g/kg DM) and the absolute amount of ADL (g) of CAU3138 from 7 days on; the content and absolute amount of hemicellulose also decreased with prolonged treatment times. The I. lacteus caused a higher loss of ADL (43 g/kg DM) in the 1-mm CAU3138 compared to the control, but it also caused a high loss of hemicellulose (106 g/kg DM). After 42 days of incubation, the ADL content of the 2-cm CAU3138 was lower (P < 0.05) than that of the 1-mm particles. However, the cellulose content of the 2-cm CAU3138 treated by the fungus was also lower than the 1-mm CAU3138 (P < 0.05). Even more notable, the absolute amount of cellulose loss was significantly larger than it was for the 1-mm CAU3138 treated with the fungus (P < 0.01). The CP content usually indicates the nutritional value of a feed and, in this study, the CP content increased significantly after the fungus treatment of CAU3138 with the prolonged treatment time (P < 0.01). However, the absolute amount of CP only increased by a small amount from the prolonged fungus treatment time. Enlarging the particle size improved the absolute amount of CP as well as the CP content for the sample treated by I. lacteus.
Table 3. Chemical Composition of 1-mm and 2-cm Corn Stover CAU3138 Over a 42-Day Period of Treatment with Irpex lacteus
Note: a to f Means of each fungus treatment within a column followed by the same letter are not significantly different from each other at P < 0.05; **- P < 0.001; *- P < 0.05; ADL- acid detergent lignin; HC- hemicelluloses; Cell- cellulose; CP- crude protein In this study, the fungal strains that grew rapidly and could inhibit mould and bacterial contamination were selected (Tirado-González et al. 2016). The strains of P. ostreatus and I. lacteus, which displayed high growth speeds, presented lower rates of mould contamination indicating that these fungi may have had bioactive compounds that acted as antimicrobials (Chaudhary and Tripathi 2015). Higher SV indicate relatively low cellulose losses during the fungal treatment process; an SV of 1.0 means that lignin and cellulose were equally lost (Zhang et al. 2007; Saha et al. 2016). There are several reports on using SV to screen for the fungi best at degrading lignocelluloses for various biofuel applications (Zhang et al. 2007; Wan and Li 2010; Saha et al. 2016). However, SV was employed in this study as an initial prediction of which fungi would be best able to degrade corn stover for the use of ruminant feed, although the results indicated that SV was not reliable to screen the fungi for improvement of the in vitro rumen digestibility of corn stover. The ADL content of CAU3138 was almost three-fold higher than that of ZN01 in the current study. The ADL is one of the main components of plant cell walls that limits rumen microorganism utilization of the cellulose and hemicellulose (Van Kuijk et al. 2015a). Therefore, the likely reason that the IVGP of CAU3138 and ZN01 treated with the same fungi showed different results was the corn stovers’ different ADL contents. Moreover, Tuyen et al. (2013) ever reported that nutritional value improvements by some fungi treatments linearly with ADL content increased in substrates. In that study, four species of white-rot fungi treatments of rice straw, maize stover, oil palm fronds, and sugarcane bagasse were compared, and it was revealed that, for a poorly lignified feedstuff, nutritional value is not improved by treatment with Ceriporiopsis subvermispora, Lentinula edodes, Pleurotus eryngii, or Pleurotus ostreatus. It was confirmed in this study that the total IVGP after fungal treatment of the (lower ADL content) corn stover ZN01 was not improved compared with the control. However, fungal treatment of the higher ADL content CAU3138 resulted in a large increase of IVGP after treatment with I. lacteus, which indicated that I. lacteus has the potential to improve the nutritional value of corn stover with higher ADL content. In addition, the higher SV of P. cystidiosus treatment but lower (P < 0.01) gas production indicated that there was no close relationship between SV and IVGP. This was inconsistent with the screening method, which was based on the SV applied in the biofuels industry, and different from the conclusions that there is a close relationship between the increase in gas production and the lignin to cellulose loss ratio for white-rot fungal treatment of wheat straw reported by Tuyen et al. (2012). However, this result was consistent with Niu et al. (2018), who reported that P. ostreatus had a higher loss ratio of ADL to cellulose than I. lacteus but also had a lower gas production. The differing results were likely due to the patterns of attack on lignocellulosic biomass by fungi are different (Wan and Li 2010), and, possibly the diverse characteristic of the substrates. The effect of the duration of the I. lacteus treatment period on IVGP (for CAU3138 stover) indicated that prolonging the treatment by this fungus could increase the IVGP. However, this might have been due to the significant decrease in ADL content between days 28 and 42 by this fungus, thus improving the availability of fermentable nutrients in the stover for rumen microbes. Tuyen et al. (2013) reported that after 6 weeks C. subvermispora and P. eryngii increased the total gas production of corn stover more than the autoclaved but un-inoculated substrates (control) and a sample with a 3-week incubation period, and that the samples treated with L. edodes and P. ostreatus had less gas production than the control. This was possibly because the ADL content of the corn stover used in that study was less than that of the CAU3138 used in the present research. The composition of gas production in vitro had been reported by Tuyen et al. (2013), who indicated that the use of C. subvermispora to improve the nutritive value of the oil palm fronds will potentially result in a decreased methane emission. Therefore, the composition of gas production in vitro in this study should be analyzed in the future to make clear whether I. lacteus treatment can decrease the methane emission. The particle size of the substrate had differing effects on the fungal treatments of the corn stovers. Salvachúa et al. (2013) reported that digestibility was significantly improved as particle size was reduced for I. lacteus-treated wheat straw cultures. This is explained by the fact that with decreasing particle sizes, a larger surface to volume ratio is obtained, which makes the carbohydrates more accessible. Gómez et al. (2011) demonstrated this when Trametes sp. was grown on smaller particles of corn stover and an expression of cellulases and xylanases was increased. However, it is not practical to apply such small particle sizes of corn stover in the ruminant feed. Moreover, Van Kuijk et al. (2016) has also reported that particle size did influence L. edodes treatments of wheat straw. The L. edodes treatment of 2-cm wheat straw resulted in a more selective delignification and a higher IVGP value than the smaller particles. Furthermore, to reach the carbohydrates in large particles, the fungi have to degrade more ADL than they would for smaller particles, which explains why the 2-cm particles CAU3138 resulted in a lower ADL after I. lacteus treatment in this study. The IVGP results of this study showed that a particle size of 2 cm had a negative influence on I. lacteus treatment of CAU3138. This was probably because the cellulose content also decreased after the fungus treated the larger particle size of the stover. However, the IVGP still increased compared with the control. Nonetheless, the effects of I. lacteus treatment on other particle sizes of CAU3138 between 1-mm and 2-cm should be further studied to optimize the appropriate particle size. The ADL content was slightly increased in the initial stage of fungal treatment of 1-mm and 2-cm CAU3138 on day 7 mainly due to the heavy loss of other components. This was similar to the wheat straw treatment results with L. edodes reported by Van Kuijk et al. (2015b). Prolonging the treatment time further degraded the ADL content of 1-mm CAU3138, which was consistent with the study by Xu et al. (2010) but would be inadvisable for the utilization of corn stover because there was too great of a loss in DM. The cellulose loss seen in the I. lacteus treatment of 2-cm CAU3138 was larger than that from the 1-mm CAU3138, which is disadvantageous for the effective utilization of the corn stover; an increase in CP content suggests an increase of nitrogen in the treated corn stover. This result needs further clarification as it is generally accepted that white-rot fungi are unable to fix atmospheric nitrogen from the air (Millbank 1969). However, the enrichment of the CP content of CAU3138 was consistent with the research of Nayan et al. (2018). The increased CP content in this study might have been at the expense of other nutrients. Moreover, the fungal mycelium protein and the liberate cell wall bound protein probably also contributed to the increase of the soluble protein.
CONCLUSIONS
Seven fungi used in this study were not able to improve the rumen degradability of corn stover with low ADL content. However, I. lacteus produced the most beneficial result in that it can enhance the rumen degradability of the corn stover with high ADL content. Therefore, this fungus can improve efficiencies in ruminant feeding in the future.
This study first time indicated that not all the white rot fungi screened by SV could effectively improve the rumen degradability of corn stover.
Longer treatment times, as well as smaller particle sizes, increased the efficiency of I. lacteus to improve the rumen degradability of high ADL content of corn stover.
ACKNOWLEDGEMENTS
The authors appreciate the financial support from the Ministry of Science and Technology of the People’s Republic of China (2015DFG32360).
REFERENCES CITED
Chaudhary, R., and Tripathi, A. (2015). “Isolation and identification of bioactive compounds from Irpex lacteus wild fleshy fungi,” J. Pharm. Sci. Res. 7(7), 424-434. http://www.jpsr.pharmainfo.in/Documents/Volumes/vol7Issue07/jpsr07071508.pdf
Chen, Y., Fan, H., and Meng, F. (2017). “Pleurotus ostreatus decreases cornstalk lignin content, potentially improving its suitability for animal feed,” J. Sci. Food Agr. 97(5), 1592-1598. DOI: 10.1002/jsfa.7907
Gómez, S. Q., Cuenca, A. A., Flores, Y. M., Rodríguez, J. N. G., and Jurado, A. T. (2011). “Effect of particle size and aeration on the biological delignification of corn straw using Trametes sp 44,” BioResources 7(1), 327-344. DOI: 10.15376/biores.7.1.327-344
Menke, K. H., Raab, L., Salewski, A., Steingass, H., Fritz, D., and Schneider, W. (1979). “The estimation of the digestibility and metabolizable energy content of ruminant feeding stuffs from the gas production when they are incubated with rumen liquor in vitro,” J. Agr. Sci. 93(1), 217-222. DOI: 10.1017/S0021859600086305
Millbank, J. W. (1969). “Nitrogen fixation in moulds and yeasts—a reappraisal,” Arch. Mikrobiol. 68(1), 32-39. DOI: 10.1007/bf00408444
Nayan, N., Sonnenberg, A., Hendriks, W. H., and Cone, J. W. (2018). “Variation in the solubilization of crude protein in wheat straw by different white-rot fungi,” Anim. Feed Sci. Tech. 242, 135-143. DOI: 10.1016/j.anifeedsci.2018.06.009
Ning, T., Wang, H., Zheng, M., Niu, D., Zuo, S., and Xu, C. (2017). “Effects of microbial enzymes on starch and hemicellulose degradation in total mixed ration silages,” Asian Austral. J. Anim. 30(2), 171-180. DOI: 10.5713/ajas.16.0046
Niu, D., Zuo, S., Jiang, D., Tian, P., Zheng, M., and Xu, C. (2018). “Treatment using white rot fungi changed the chemical composition of wheat straw and enhanced digestion by rumen microbiota in vitro,” Anim. Feed Sci. Tech. 237, 46-54. DOI: 10.1016/j.anifeedsci.2018.01.005
Rahman, M. M., Lourenço, M., Hassim, H., Baars, J. J., Sonnenberg, A. S., Cone, J. W., De Boever, J., and Fievez, V. (2011). “Improving ruminal degradability of oil palm fronds using white rot fungi,” Anim. Feed Sci. Tech. 169(3-4), 157-166. DOI: 10.1016/j.anifeedsci.2011.06.014
Saha, B. C., Qureshi, N., Kennedy, G. J., and Cotta, M. A. (2016). “Biological pretreatment of corn stover with white-rot fungus for improved enzymatic hydrolysis,” Int. Biodeter. Biodegr. 109, 29-35. DOI: 10.1016/j.ibiod.2015.12.020
Salvachúa, D., Prieto, A., López-Abelairas, M., Lu-Chau, T., Martínez, Á. T., and Martínez, M. J. (2011). “Fungal pretreatment: An alternative in second-generation ethanol from wheat straw,” Bioresource Technol. 102(16), 7500-7506. DOI: 10.1016/j.biortech.2011.05.027
Salvachúa, D., Prieto, A., Vaquero, M. E., Martinez, A. T., and Martinez, M. J. (2013). “Sugar recoveries from wheat straw following treatments with the fungus Irpex lacteus,” Bioresource Technol. 131, 218-225. DOI: 10.1016/j.biortech.2012.11.089
Syed, K., and Yadav, J. S. (2012). “P450 monooxygenases (P450ome) of the model white rot fungus Phanerochaete chrysosporium,” Crit. Rev. Microbiol. 38(4), 339-363. DOI: 10.3109/1040841x.2012.682050
Tirado-González, D. N., Jáuregui-Rincón, J., Tirado-Estrada, G. G., Martínez-Hernández, P. A., Guevara-Lara, F., and Miranda-Romero, L. A. (2016). “Production of cellulases and xylanases by white-rot fungi cultured in corn stover media for ruminant feed applications,” Anim. Feed Sci. Tech. 221(Part A), 147-156. DOI: 10.1016/j.anifeedsci.2016.09.001
Tuyen, D. V., Phuong, H. N., Cone, J. W., Baars, J. J., Sonnenberg, A. S., and Hendriks, W. H. (2013). “Effect of fungal treatments of fibrous agricultural by-products on chemical composition and in vitro rumen fermentation and methane production,” Bioresource Technol. 129, 256-263. DOI: 10.1016/j.biortech.2012.10.128
Tuyen, V. D., Cone, J. W., Baars, J. J., Sonnenberg, A. S., and Hendriks, W. H. (2012). “Fungal strain and incubation period affect chemical composition and nutrient availability of wheat straw for rumen fermentation,” Bioresource Technol. 111, 336-342. DOI: 10.1016/j.biortech.2012.02.001
Van Kuijk, S. J., Sonnenberg, A. S., Baars, J. J., Hendriks, W. H., and Cone, J. W. (2016). “The effect of particle size and amount of inoculum on fungal treatment of wheat straw and wood chips,” J. Anim. Sci. Biotechno. 7, 39-47. DOI: 10.1186/s40104-016-0098-4
Van Kuijk, S. J. A., Sonnenberg, A. S. M., Baars, J. J. P., Hendriks, W. H., and Cone, J. W. (2015a). “Fungal treated lignocellulosic biomass as ruminant feed ingredient: A review,” Biotechnol. Adv. 33(1), 191-202. DOI: 10.1016/j.biotechadv.2014.10.014
Van Kuijk, S. J. A., Sonnenberg, A. S. M., Baars, J. J. P., Hendriks, W. H., and Cone, J. W. (2015b). “Fungal treatment of lignocellulosic biomass: Importance of fungal species, colonization and time on chemical composition and in vitro rumen degradability,” Anim. Feed Sci. Tech. 209, 40-50. DOI: 10.1016/j.anifeedsci.2015.07.026
Van Soest, P. J. (1973). “Collaborative study of acid-detergent fiber and lignin,” Journal of the AOAC. 56(4), 781-784.
Van Soest, P. J.., Robertson, J., and Lewis, B. (1991). “Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition,” J. Dairy Sci. 74(10), 3583-3597. DOI: 10.3168/jds.s0022-0302(91)78551-2
Wan, C., and Li, Y. (2010). “Microbial delignification of corn stover by Ceriporiopsis subvermispora for improving cellulose digestibility,” Enzyme. Microb. Tech. 47(1-2), 31-36. DOI: 10.1016/j.enzmictec.2010.04.001
Xu, C., Ma, F., Zhang, X., and Chen, S. (2010). “Biological pretreatment of corn stover by Irpex lacteus for enzymatic hydrolysis,” J. Agr. Food. Chem. 58(20), 10893-10898. DOI: 10.1021/jf1021187
Zhang, X., Yu, H., Huang, H., and Liu, Y. (2007). “Evaluation of biological pretreatment with white rot fungi for the enzymatic hydrolysis of bamboo culms,” Int. Biodeter. Biodegr. 60(3), 159-164. DOI: 10.1016/j.ibiod.2007.02.003
Zuo, S., Niu, D., Zheng, M., Jiang, D., Tian, P., Li, R., and Xu, C. (2018). “Effect of Irpex lacteus, Pleurotus ostreatus and Pleurotus cystidiosus pretreatment of corn stover on its improvement of the in vitro rumen fermentation,” J. Sci. Food. Agr. 98(11), 4287-4295. DOI: 10.1002/jsfa.8951
Article submitted: October 22, 2018; Peer review completed: November 26, 2018; Revisions accepted: December 7, 2018; Published: December 11, 2018.
DOI: 10.15376/biores.14.1.895-907
