Abstract
Marine angiosperms could inevitably offer considerable potential resources for their fiber, yet little research has been conducted, especially in Malaysia. Fiber characteristics of five species of seagrass – Enhalus acoroides, Cymodocea serrulata, Thalassia hemprichii, Halophila ovalis, and Halophila spinulosa – were evaluated. Fiber dimensions were studied to determine slenderness ratio, flexibility coefficient, Runkel’s ratio, and Luce’s shape factor species selection. The seagrass species have the potential in papermaking production as they possessed slenderness ratio >33 (98.12 to 154.08) and high Luce’s shape factor (0.77 to 0.83); however the species exhibited low flexibility coefficient <50 (30.07 to 35.18) and >1 Runkel’s ratio (1.11 to 1.60), which indicate rigid fiber. The five seagrass species have high cellulose >34% (40.30 to 77.18%) and low lignin content <15% (5.02 to 11.20%), which are similar to those encountered in non-wood plant species. Handmade paper sheet of Enhalus acoroides using pulp subjected to mechanical blending exhibited the highest tensile strength (4.16 kN/m) compared to hand-beaten pulp (3.46 kN/m). The highest breaking length (3.43 km) was achieved by a paper sheet of Thalassia hemprichii using hand-beaten pulp. Based on their physical and chemical composition properties, seagrass have potential as sources of fibrous material for handmade papermaking.
Download PDF
Full Article
Fiber Characteristics and Papermaking of Seagrass Using Hand-beaten and Blended Pulp
Nurul Nur Farahin Syed,a Muta Harah Zakaria,a,b,* and Japar Sidik Bujang c
Marine angiosperms could inevitably offer considerable potential resources for their fiber, yet little research has been conducted, especially in Malaysia. Fiber characteristics of five species of seagrass – Enhalus acoroides, Cymodocea serrulata, Thalassia hemprichii, Halophila ovalis, and Halophila spinulosa – were evaluated. Fiber dimensions were studied to determine slenderness ratio, flexibility coefficient, Runkel’s ratio, and Luce’s shape factor species selection. The seagrass species have the potential in papermaking production as they possessed slenderness ratio >33 (98.12 to 154.08) and high Luce’s shape factor (0.77 to 0.83); however the species exhibited low flexibility coefficient <50 (30.07 to 35.18) and >1 Runkel’s ratio (1.11 to 1.60), which indicate rigid fiber. The five seagrass species have high cellulose >34% (40.30 to 77.18%) and low lignin content <15% (5.02 to 11.20%), which are similar to those encountered in non-wood plant species. Handmade paper sheet of Enhalus acoroides using pulp subjected to mechanical blending exhibited the highest tensile strength (4.16 kN/m) compared to hand-beaten pulp (3.46 kN/m). The highest breaking length (3.43 km) was achieved by a paper sheet of Thalassia hemprichii using hand-beaten pulp. Based on their physical and chemical composition properties, seagrass have potential as sources of fibrous material for handmade papermaking.
Keywords: Seagrass; Fiber dimension; Chemical composition; Papermaking; Handmade paper; Mechanical strength
Contact information: a: Department of Aquaculture, Faculty of Agriculture, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor Darul Ehsan, Malaysia; b: Institute of Bioscience, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor Darul Ehsan, Malaysia; c: Department of Biology, Faculty of Science, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor Darul Ehsan, Malaysia;
* Corresponding author: muta@upm.edu.my
INTRODUCTION
In ancient times, humans used non-wood plants as surfaces for writing. Cyperus papyrus L. was widely used as writing material by Egyptians since 3000 BC (Saijonkari-Pahkala 2008). The first recorded practitioner of the papermaking technique, Ts’ai Lun, also used non-wood plants, such as hemp, rags, and mulberry to produce paper sheets. There is an increasing trend of pulp production from non-wood sources worldwide, e.g., China and India, mainly because of the shortage of hardwood fiber raw material (Oinonen and Koskivirta 1999; Saijonkari-Pahkala 2008). Han James (1998) reported that certain non-wood plants such as Gossypium sp. have high cellulose content (85% to 90%) compared to the wood of Pinus sylvestris (35% to 49% cellulose). Hemp, Cannabis sp. also has a low lignin content (3%), which suggests that these non-wood fibers are suitable for papermaking. Photosynthetic aquatic species such as seagrass also contain cellulose or other fibrous materials potentially suitable for paper production, with the added advantage of faster propagation and maturation than terrestrial plants.
Fiber characteristics vary between plants, and analysis of physical properties and chemical composition of the fiber is important in order to provide an indication of the papermaking potential of various species (Saijonkari-Pahkala 2008). Fiber morphology such as fiber length and width are important in determining pulp fibers quality (Wood 1981). One of the most important physical properties of fibers for papermaking is fiber length, which basically influences the tearing strength of the paper (Marques et al. 2010). Plant fibers consist of three structural polymers, which are the polysaccharides cellulose and hemicellulose, plus the aromatic polymer lignin, as well as some minor non-structural components such as proteins, extractives, and minerals (Marques et al. 2010). Some non-wood fibers contain more pentosans (>20%), holocellulose (>70%), and less lignin (≃15%) compared to hardwoods (Hunsigi 1989). In addition, the higher hot water solubility characteristic possessed by non-wood plants ease the cooking liquors accessibility (Saijonkari-Pahkala 2008).
Photosynthetic aquatic species such as seagrass also contains cellulose or other fibrous materials potentially suitable for paper production. Seagrasses are aquatic angiosperms, which are restricted to the marine environment (Kuo and den Hartog 2001). In Malaysia, the seagrass can be found mostly in the shallow inter-tidal, semi-enclosed lagoons, sub-tidal zones, coral reef, and mangrove ecosystems (Japar Sidik and Muta Harah 2003). Peoples in Ria de Aveiro have been collecting more than 100,000 tons of aquatic vegetation (e.g., Potamogeton pectinatus, Ruppia cirrhosa, and Zostera noltii) per year including seagrass (Silva et al. 2004; Cunha et al. 2013). In Germany, seagrass fiber was substituted for cotton in the manufacture of nitrocellulose during the Second World War, (Milchakova et al. 2014). Based on the above information, although seagrass contain fiber which can be used for papermaking, the pulping method suitability as raw materials for papermaking were not fully explored during the 1980s (Cunning 1989). With this prospective, an attempt was made to evaluate the suitability of seagrasses in papermaking based on their fiber morphology and chemical composition. Tested parameters on the paper properties including tensile strength, breaking length, stress-strain curve, and modulus elasticity were compared and recorded based on seagrass species fibers and pulping method.
EXPERIMENTAL
Raw Materials
Enhalus acoroides, Thalassia hemprichii, Cymodocea serrulata, Halophila ovalis and Halophila spinulosa were collected along the sub tidal shoals of Tanjung Adang-Merambong shoals (01° 19’ N, 103° 36’ E). Entire seagrass plants were collected, except for Enhalus acoroides, where only the blades were used as raw materials for handmade papermaking. The samples were cleaned and placed in an ice chest before being transported to the laboratory for further processing as described below.
Physical Properties Identification
Cleaned fresh plants were fragmented into small pieces (2 cm) and macerated in 10 ml of 67% nitric acid (HNO3) in a test tube and boiled in a water bath at 100±2 °C for 10 min. The samples were then washed with over flowing tap water before placing in a centrifuge tube containing 10 mL of distilled water (Ververis et al. 2004). The macerated fiber suspensions in the tube was mixed using vortex before being examined under calibrated compound microscope, ZEISS Germany Axioskop attached with camera Nikon model DS-Fi1 to measure the fiber length (L), fiber diameter (D), lumen diameter (d), and cell wall thickness (W) (Fig. 1). The derived values; slenderness ratio (L/D), Runkel’s ratio (2W/d), flexibility ratio [d/(D x 100)], and Luce’s shape factor [(D2–d2)/(D2+d2)] of each fiber dimensions were calculated in order to assess the fiber quality for paper production (Ohshima et al. 2005).

Fig. 1. Fiber wall of seagrass (20X magnification). (a) Thin walled Halophila spinulosa fiber (b) Thick walled Enhalus acoroides fiber. LD represents lumen diameter.
Chemical Composition Identification
Air dried seagrass plants were ground and passed through a 70 µm mesh screen. Direct estimation of cellulose, hemicellulose, and lignin were carried out using the method as described by Moubasher et al. (1982). Two grams of ground sample were boiled in 2:1 ethanol-toluene solvent for 4 h using Soxhlet extraction and washed thoroughly and dried (105 °C) in an air circulation oven overnight. The dry samples were weighed and divided into 2 parts. The first part was labeled as fraction A, while the second part was treated with 24% potassium hydroxide (KOH) solution for 4 h at 25 °C, then filtered using a weighed glass extraction thimble, labeled as fraction B. The fraction B was further treated with 72% sulphuric acid (H2SO4) for 3 h and filtered using the glass thimble and refluxed with 5% H2SO4 at 90 °C for 2 h. The residues were then filtered and washed to remove the H2SO4 and later dried at 80 °C for 24 h. The dry samples were weighed as fraction C.
Cellulose content = B – C
Hemicellulose content = A – B
Lignin content = C
Pulp and Papermaking
Dried seagrass plants were chopped into small fragments (2 to 5 cm length) and completely soaked in water overnight to soften the fibers. Samples were cooked in a solution having the ratio of 1 water to 2 sodium hydroxide (NaOH) using an ELBA induction cooker at a constant temperature of 160 °C for 2 to 6 h as shown in Table 1. The cooking durations varied with seagrass plant species; for example Enhalus acoroides need longer cooked time and with additional water and NaOH for the pulp to soften. Fibers were then washed with water to remove the residue.
Each pulp derived from seagrass plant species were processed using two different methods; (i) beaten using wooden mallet for 20 min until the fiber spread out and became soft, (ii) and blended for 10 min. The next stage was sheet formation, in which the processed pulps were diluted with water in a vat using mould and deckle dipped them under the fibers. The deckles containing the pulp were lifted, shaken slightly to help distribute the fibers evenly, before being laid down on the felt, and the deckle was carefully removed. Excessed water was removed by air drying at room temperature (25 to 27 °C), pressed, and oven dried at 60 °C for 3 days.
Table 1. Yield and Pulp Weight of Seagrass during Cooking Process
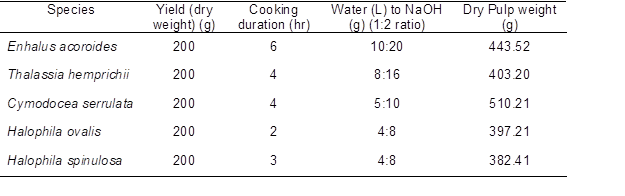
Mechanical Strength of Paper
Fiber distribution of paper sheets produced was observed under ZEISS Stemi SVII with camera PixeLINK model PL-A662. Tensile strength of each paper was measured by following TAPPI Method T-404 (1992). The handmade paper was cut into 5 strips, each with a dimension of 1 cm wide x 5 cm long. The strips were weighed, and the thickness was measured using the micrometer (Mitutoyo, Japan) before being tested using Universal Testing Machine ISNTRON 3365 (5kN) at the rate of elongation of 5 mm per minute. All the data (tensile stress, strain and modulus elasticity) were recorded using the Bluehill® software. The tensile strength and breaking length were calculated using the formula as followed:
Tensile strength (kN/m) = maximum breaking force (kN)/ paper width (m)
Breaking length (km) = 120,000 (tensile strength/ grammage)
Statistical Analysis
Comparison for fiber dimension, derived values and chemical composition between seagrass species were using 1-way ANOVA and if result is significant, this was followed by a post-hoc Tukey’s Test (p<0.05) using Statistical Package for the Social Science (SPSS) software. Data comparison for paper produced using hand-beaten and blended pulp were analyzed using Independent Sample T-Test (p<0.05). Principal component analysis (PCA) based on the Pearson method was carried out using XLSTAT software version 2013 (Addinsoft, New York, USA) to obtain the relationship between variables obtained for seagrass species compared with other non-wood plant species from other studies.
RESULTS AND DISCUSSION
Fiber Dimensions and Derived Valued
Table 2 shows the fiber dimensions of seagrass. Fiber length of Enhalus acoroides (2.04 mm) was significantly longer (p<0.05) compared with other species (ranged 1.51 to 1.55 mm). Based on the standard published by International Association of Wood Anatomy (IAWA 1937), among the species tested, Enhalus acoroides is categorized as moderately long fiber (1600 to 2200 µm), while the rest as medium-sized fiber (1200 to 1500 μm).
The strong fibers of Enhalus plants are also being utilized for construction of nets by people in Yap Island (Falanruw 1992). Longer fiber tends to be less uniformly distributed in a paper sheet and can cause malformation. The proportion of fiber length to diameter which is suitable for paper production is about 100:1, while for textiles a typical fiber length is >1000:1 (Saijonkari-Pahkala 2008). In this study, Enhalus acoroides with 128:1 ratio indicated its suitability in papermaking, and the value is slightly higher compared to 125:1 for Cyperus papyrus (Hurter 1990; Saijonkari-Pahkala 2008). In contrast, Halophila ovalis has wider fiber diameter (20.87 µm), lumen diameter (6.91 µm), and cell wall thickness (7.81 µm) compared to others. Lumen size is important for fiber dimensions because it will effect on the rigidity and strength of paper (Akpakpan et al. 2012). However, greater value of fiber diameter increases the void volume and forms a coarse-surfaced paper sheet (Kaur and Dutt 2013). Large fibers with thin walls also give a positive effect as they tend to form non-porous tightly bonded paper sheet that is easily collapse and flexible. In contrast, the less flexible thick walled fiber will produced less bounded and bulkier paper sheet resulting in a lower burst (Akpakpan et al. 2012).
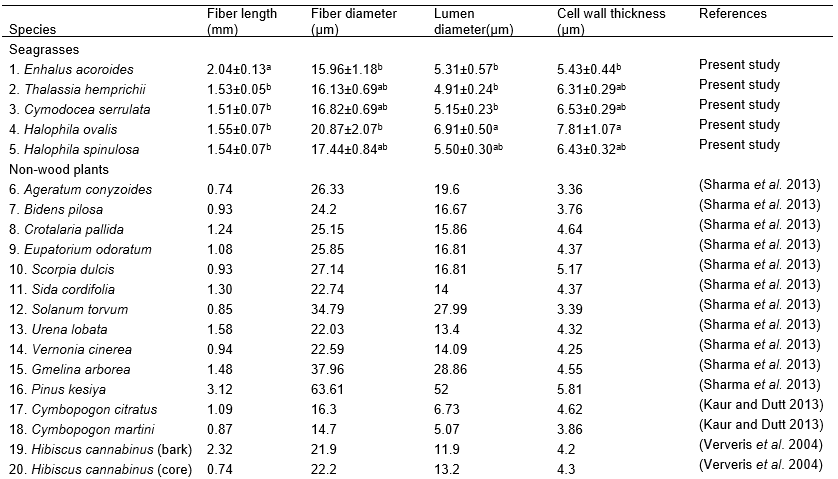
Derived values from the fiber dimensions are important to determine the suitability of material for paper production. There were significant differences in slenderness ratio values (p<0.05) between Enhalus acoroides (154.08) with the rest of seagrass species (Table 3). It is invaluable for pulp and paper quality if the slenderness ratio of fibrous material is <70 (Young 1981). According to Sharma et al. (2013) and Xu et al. (2006) slenderness ratio of >33 is considered good for pulp and paper production. All the seagrass species in this study exceeded the satisfactory slenderness ratios of 33. A higher slenderness ratio in fibers will produce higher rate of paper tear resistance (Akpakpan et al. 2012). Based on PCA, the seagrass species and other non-woods plants were clustered into five main groups (Fig. 2). The first two factors (F) accounted for 86.55% of total variance. F1 explained the high percentage of total variance (66.21%) compared to F2 (20.34%). Fiber diameter, lumen diameter, and flexibility coefficient were positively correlated to F1, while fiber length, cell wall thickness, slenderness ratio, and Runkel ratio were positively correlated to F2. Based on their slenderness ratio, all the seagrass species were in Group 2, clustered with Kenaf bark (Hibiscus cannabinus). Kenaf bark comprises 35% of fibrous part (Manzanares et al. 1997) and contains two fiber components that are different in morphology and chemical properties, and both are suitable for paper and paperboards (Shakhes et al. 2011; Touzinski et al. 1973), writing, printing, packaging and wrapping purposes (Ververis et al. 2004). Group 1 have the highest number of non-wood plants, which shared similar value of fiber diameter, lumen diameter, and flexibility coefficient. The members of Group 3, which comprised grass, lemon grass, and cane, were clustered together, as they have high value of Runkel ratio. Pinus kesiya and Gmelina arborea were together in Group 4, having a high value of lumen diameter. Camellia sinensis plant had the highest value in all fiber morphology variables and was clustered in Group 5.
Table 2. Fiber Morphology of Seagrass and Other Non-wood Plants

There are four groups of fibers based on the flexibility coefficient (Bektas and Tutus 1999; Hemmasi et al. 2011); (a) high elastic fibers with elasticity ratio >75, (b) elastic fibers with elasticity ratio between 50 and 75, (c) rigid fibers with elasticity ratio between 30 and 75, and (d) highly rigid fibers with ratio <30. In this present study all the seagrass have rigid fiber with flexibility coefficient (30.07 to 35.18). As the flexibility coefficient increase, the suppleness of the fibers also increase, resulting in a well-bonded sheet (Akpakpan et al. 2012). During the papermaking process, the rigid fibers will not conform in the paper sheet, hence decreasing the fiber bonding and elasticity. Rigid fibers are usually used more on fiber plate, rigid cardboard, and cardboard production (Akgul and Tozluoglu 2009).
All seagrass species in this present study have Runkel’s ratio >1, which corresponds to their rigid fiber characteristic. Eroglu (1980) has classified the most suitable Runkel’s ratio as <1 where the cell wall is thin as the fibers are more flexible and form paper with large bonding. Runkel’s ratio equal to 1 is also suitable for paper production but less preferable. The fiber properties with this Runkel value is hard and stiff, resulting in poor bonding ability and therefore reduced paper quality (Wahab et al. 2006). This value can be improved by using a suitable screening technique which needs more cost (Ververis et al. 2004).
The Luce’s shape factor indices did not vary to an important degree between the five seagrass species (0.77 to 0.83) and comparable to the reed species (Arundo donax). This showed that the mechanical and structural properties of seagrass plants were in range with reed, compared to the other non-wood species (Table 3). The Luce’s shape factor is derived from circular shape of both fiber diameter and fiber lumen diameter, which give the indicator to paper sheet density and the breaking length of paper (Ona et al. 2001).
Chemical Composition
Chemical composition of all species showed good percentage value (40.30 to 77.18%) for cellulose, which is the main component of plant fibers used during pulping (Table 4). Enhalus acoroides has the highest cellulose content with 77.18% (p<0.05) compared to others. According to Nieschlag (1960), plant fibers with cellulose >34% would be preferable for pulp and paper manufacture. Higher cellulose content of plant material will provide stability and high tensile strength for the paper produced (Agu et al. 2012). Enhalus acoroides and Zostera marina were clustered in Group 4 (Fig. 3) due to similarity in hemicellulose and lignin content. Good mechanical properties of marine eelgrass, Z. marina fibers which contain high polysaccharides and pectin but low lignin are comparable to sisal and jute (Davies et al. 2007). Other previous study showed that Z. marina and green algae, Cladophora sp. produce a similar strength as ground wood, however due to its low brightness, these plants are only useful as filler material up to 20% in papermaking (Knoshaug et al. 2009; Leopold and Marton 1974).
The hemicellulose content of the seagrass ranged between the lowest (14.33%) in Thalassia hemprichii to the highest (28.31%) in Enhalus acoroides. This value is within the range of wheat straw (23 to 30), a widely used non-wood plants papermaking in China (Ogunwusi 2014). Higher hemicellulose values result in higher strength of paper (especially tensile, burst, and fold) and the yield of pulp (Biermann 1996); however, it may have a negative effect during pulping, because hemicellulose is the cell wall polymer component having the highest water sorption (Madsen et al. 2004).
Table 3. Derived Value of Seagrass and Other Non-wood Plants
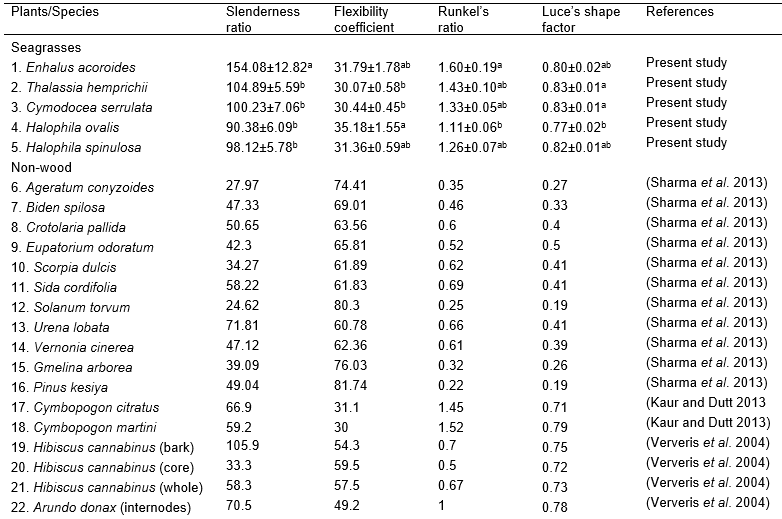
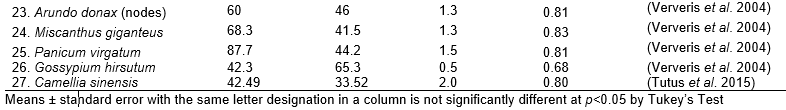
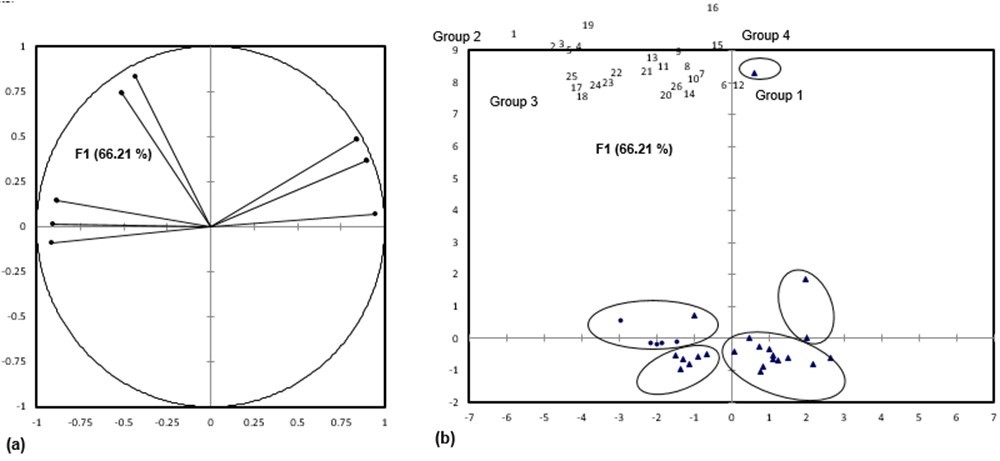
Fig. 2. (a) Plot of fiber characteristics (fiber length, fiber diameter, lumen diameter, cell wall thickness, slenderness ratio, Runkel’s ratio, Luce’s shape factor, and flexibility coefficient). (b) Position of PCA score of seagrass species (●) and other non-wood plants (▲)
A low content of hemicellulose decreases the capacity of water absorbing (Bakker and Elbersen 2005), thus minimizing the duration of pulping activity. A review by Knoshaug et al. (2013) reported that hemicellulose is not typically required and as useful as wet end additives or fillers; thus levels in the range 30 to 50% hemicellulose of the biomass materials would be adequate in papermaking using aquatic species.
Thalassia hemprichii possessed relatively low lignin content (5.02%), while a higher content was observed in Cymodocea serrulata (11.20%). The pulp and paper industry would prefer low lignin (<15%) content because the fibers are more porous and flexible and they give a brighter colour during bleaching. The PCA in Fig. 3 showed that the two factors accounted for 92.26% of total variance, with high percentage of F1 (60.67%) compared to F2 (31.59%). Three seagrass species Cymodocea serrulata, Halophila ovalis, and Halophila spinulosa were clustered with reed canary grass (Phalaris arundinacea) and banana (Musa sp.) pseudo-stem in Group 2 based on their lignin content. As reported in banana pseudo-stem, high cellulose and low lignin content give a high potential in papermaking, boards and composite materials production (Cordeiro et al. 2004). Enhalus acoroides shared a similar chemical composition with Zostera marina (Group 4) which studied to be particularly suitable for use in bio-degradable structures. Thalassia hemprichii in the present study was clustered together with Halophila ovalis and Posidonia australis (Group 3) due to their low cellulose and hemicellulose content.
Posidonia oceanica was clustered in Group 1 with 11 non-wood plants that possessed moderate values of all the variables. Thalassia testudinum was clustered in Group 5 as this seagrass was reported to have the highest content of cellulose (86.22%) and hemicellulose (65.13%).
Paper Characteristics and Mechanical Properties
The pulp from long fibers of Enhalus acoroides with higher cellulose content produced more paper sheets when compared to other species. The distribution and formation of fiber was hard to observe under the microscope in some of the produced paper sheets due to the thickness. Pressing techniques during the papermaking process also reduced the paper thickness, in which sufficient energy was needed to remove the remaining free water as much as possible in order to obtain fine paper sheets. Sirvio and Nurminen (2004) reported that, as the density is constant, the fiber network thickness is a linear function of network grammage. Thus, these three factors influenced each other, as the thickness increased, paper sheet weight for a given area increased as well. The grammage value >120 g/m2 was recorded in E. acoroides, T. hemprichii, and H. ovalis, indicating that all these species are within the range of paperboard standard (Table 5). The thinner paper sheet produced from C. serrulata and H. spinulosa resulted in lower grammage. No specific values of grammage were targeted in this work. Fiber fractionation processes can reduce the grammage variations of the paper in order to increase paper strength (Nazhad et al. 2000), cut costs, and achieve savings in raw materials (Sood et al. 2005).
Based on Fig. 4, the fiber of blended and hand-beaten can be distinguished based on their fiber network. Fibers strands can be seen on most of the blended paper (Fig. 4d-f), while fiber bundle were detected on hand-beaten paper (Fig. 4a-c). The longer beating process was required in order to improve the fiber bonding ability to shorten and formed fine fibers (Jahan and Rawshan 2009).
Table 4. Chemical Composition of Seagrass and Other Non-wood Plants
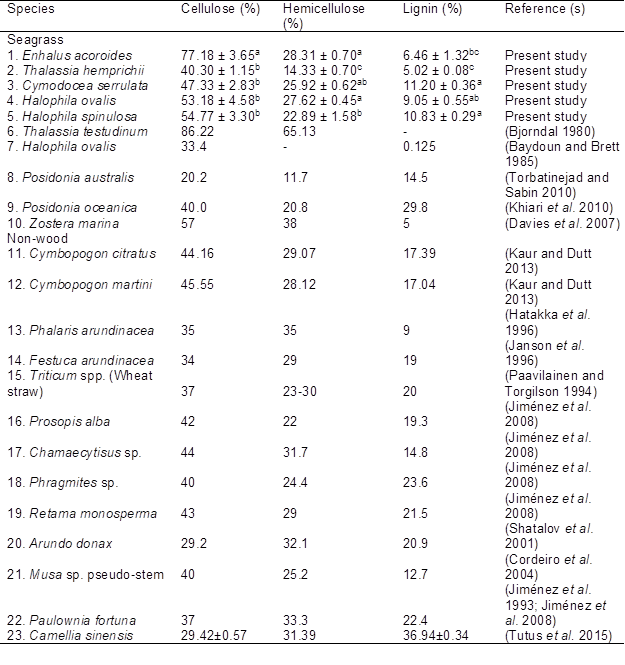
Means ± standard error with the same letter designation in the column is not significantly different at p<0.05 by Tukey’s Test
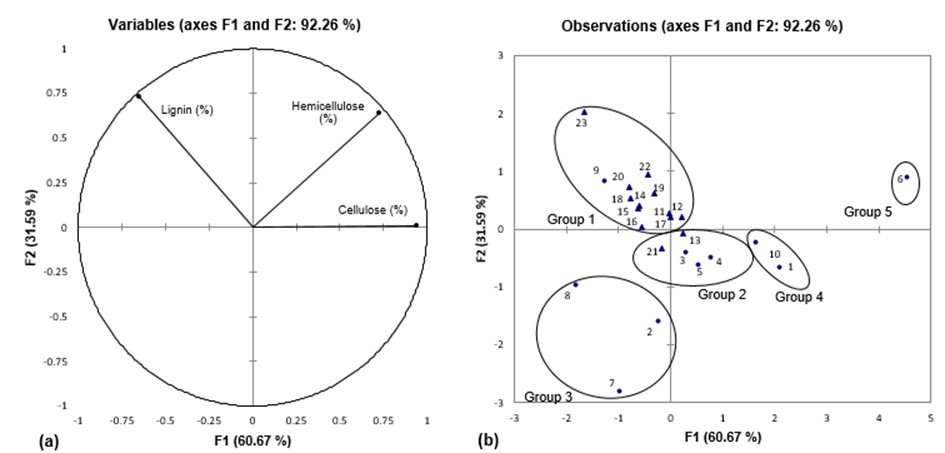
Fig. 3. (a) Plot of chemical composition (cellulose, hemicellulose and lignin). (b) Position of PCA score of seagrass species (●) and other non-wood plants (▲)

Fig 4. Fiber distribution of the seagrass paper using hand-beaten (a – c) and blended pulp (d – f). (a) Enhalus acoroides with 4.0 magnification, (b) Halophila spinulosa with 4.0 magnification, (c) Halophila ovalis with 3.2 magnification, (d) Enhalus acoroides with 3.2 magnification, (e) Halophila spinulosa with 3.2 magnification and (f) Halophila ovalis with 3.2 magnification
Table 5. Paper Sheet Properties from Seagrass Species
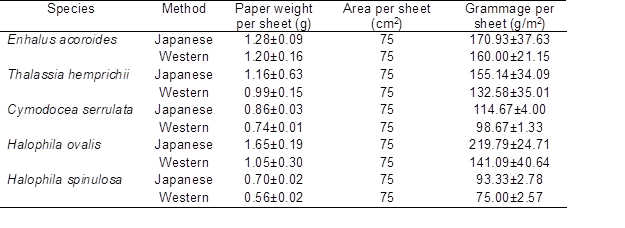
Studies showed that good fiber distributions are difficult to obtain using a hand-beating process, which requires a specific technique practiced by traditional Japanese papermakers (Barrett 1983). This explained why paper produced using blended pulp tended to have a fine fiber resulting in smoother surface (Fig. 4e) compared to the hand-beaten pulp. Flocculation was also detected, meaning that there was a non-uniform fiber distribution at some areas of the paper (Fig. 4f). Flocculation can be reduced by using shortened fibers, but this will affect the paper strength, because shorter fiber length will decrease tensile strength (Biermann 1996).
Typical stress-strain curves of paper sheets of different seagrass species by using hand-beaten and blended pulp are shown in Figs. 5 and 6, respectively. For the blended pulp paper, the highest tensile stress was 283.8 MPa from Enhalus acoroides with a strain of 0.35, where the fibers can support heavy load before being ruptured compared to the others seagrass fibers. Halophila spinulosa had the lowest tensile stress of 67.43 MPa and managed to hold a tensile strain of 0.02 when compared to Halophila ovalis (0.01).
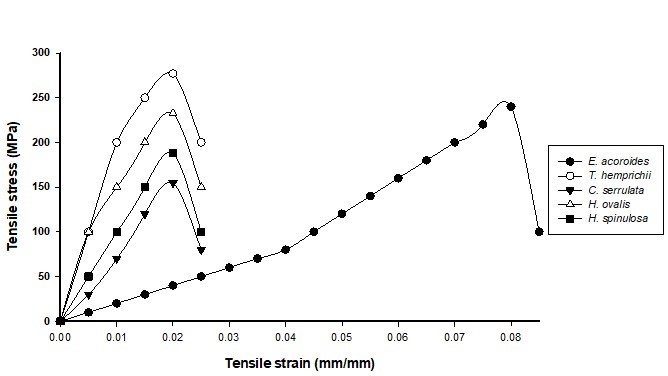
Fig. 5. Stress-strain curve of paper using hand-beaten pulp
For the hand-beaten paper, Thalassia hemprichii depicted the highest tensile stress of 277 MPa, and tensile strain of 0.02 followed by Enhalus acoroides, 240 MPa with tensile strain of 0.08. The stress increased with the strain until it attained its maximum value before the paper starts to tear apart. All four species except Enhalus acoroides have similar elongation before ruptured.
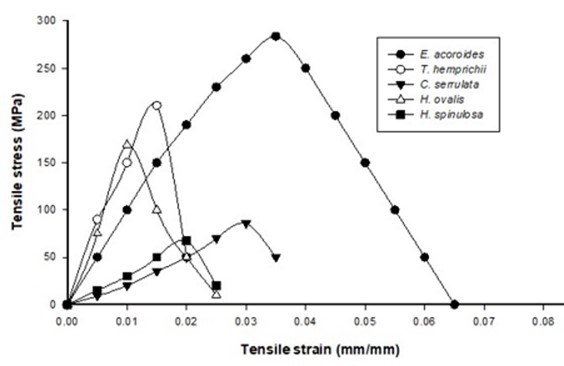
Fig. 6. Stress-strain curve of paper using blended pulp
Young’s modulus or modulus of elasticity is a measure of stiffness when the fibers are stretched or compressed. Hand-beaten paper of Enhalus acoroides possessed the highest elasticity (502.9 MPa) compared to the paper made by blended method (Fig. 7).
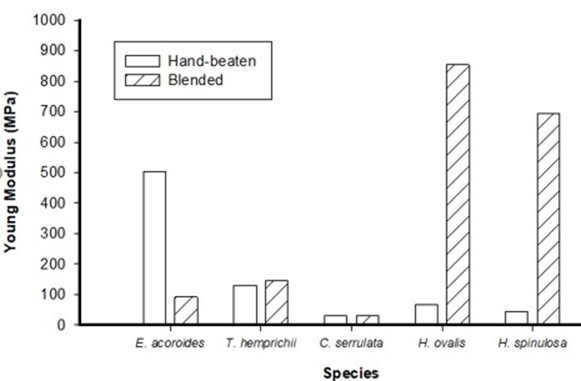
Fig 7. Young’s modulus for paper of different seagrass species using hand-beaten and blended pulp
The blended pulp of Halophila ovalis produced the highest modulus elasticity paper of 854.24 MPa due to its high fiber diameter, followed by Halophila spinulosa with 692.93 MPa. Thick wall fiber is more advantageous in tearing strength as it is stronger, flexible, and does not flatten during sheet forming compared to thin wall fiber. The higher fiber stiffness will cause the fiber to be in contact with their neighbors in the thickness direction instead of bending (Alava and Niskanen 2006).
An independent samples t-test was conducted to compare the mechanical effect of paper between hand-beaten and blended pulp. The highest tensile strength was blended pulp of Enhalus acoroides (4.10 kN/m, Table 6), while hand-beaten pulp of Thalassia hemprichii had the highest breaking length (3.43 km, Table 7). The tensile strength, fracture toughness, and breaking strain of paper were influenced by fiber length (Wathen 2006). As the fiber length increased together with high hemicellulose (Spiegelberg 1966), the tensile strength also increased which explained highest tensile strength in Enhalus acoroides. Biermann (1996) stated that typical paper has a breaking length in the range of 2.5 to 12 km, and it can be increased by the addition of a certain amount of starch (Shen et al. 2012) during the pulping process. A previous study on red algae, Gelidium corneum (2.29 kN/m, 3.84 km) and Gelidium amansii (1.86 kN/m, 3.01 km) indicated comparable tensile strength and breaking length value, respectively, with seagrass species of this present study. Thus, both materials may be suitable for many types of paper such as printing paper, filter paper, and edible paper (Seo et al. 2010).
Based on Table 5, there were significant differences when comparing tensile strength in hand-beaten pulp (M= 2.24, SD= 0.23) and blended pulp (M= 1.24, SD= 0.7) of Cymodocea serrulata, t(8)= 3.04, p= 0.016. A highly significant difference was detected in hand-beaten pulp (M= 2.22, SD= 0.5) and blended pulp (M= 0.802, SD= 0.414) of Halophila spinulosa, t(8)= 4.901, p= 0.001. High beating of pulp can improve the mechanical strength of paper (Seo et al. 2010). For breaking length (Table 6), there was a highly significant difference in beaten pulp (M= 2.43, SD= 0.22) and blended pulp (M= 1.09, SD= 0.54) of Halophila spinulosa, t(8)= 4.068, p= 0.004. Blending only cut the fiber into small fragments, while beating caused changes in fiber simultaneously including fibrillation, fiber shortening, or cutting and fines formation that improved the fiber’s bonding ability (Kang and Paulapuro 2006).
Table 6. Tensile Strength of Paper Strips of the Seagrass Species Using Two Different Methods
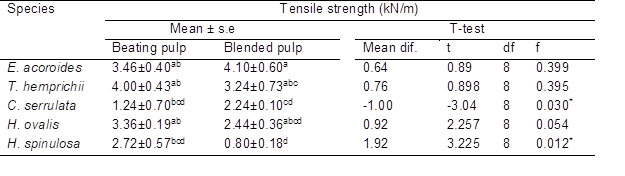
Mean ± standard error in a column with different superscript (a>b>c>d) are significantly different (p<0.05 Tukey multiple range test) * Significant at level p<0.05 for t-test
Table 7. Breaking Length of Paper Strips of the Seagrass Species Using Two Different Methods
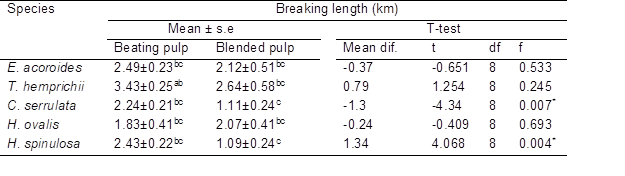
Mean ± standard error in a column with different superscript (a>b>c>d) are significantly different (p<0.05 Tukey multiple range test) * Significant at level p<0.05 for t-test
CONCLUSIONS
- This study showed that seagrass plant species’ fibers can be considered as raw materials in handmade papermaking production as they possessed moderate long (g., Enhalus acoroides) to medium-sized fibers (T. hemprichii, C. serrulata, H. ovalis and H. spinulosa), good slenderness ratio (>33), great value of Luce’s shape factor, higher cellulose (>35%), and less than 15% lignin content.
- The derived low flexibility coefficient and Runkel’s ratio resulted in rigid fibers, where seagrasses have more potential for paperboard and paper plate production.
- Of the seagrass species assessed, Enhalus acoroides is the most preferable seagrass for papermaking, as it possesses the longest fiber and highest cellulose content compared to other species.
- The low lignin content of the seagrass gives an advantage in paper strength and also in reducing the bleaching cost.
- Fiber strands produced by beating and blending required longer duration in order to obtain better fiber formation and distribution in produced paper sheets.
- It is suggested that to obtain the uniform paper sheet thickness, the grammage value must be fixed prior to paper production.
- Tensile strength and breaking length of the produced paper showed that beating method resulted in more flexible fiber compared to the blending method hence increasing the bonding strength.
ACKNOWLEDGMENTS
We would like to thank Vice-Chancellor, Universiti Putra Malaysia, for encouragement and facilities provided. This research was funded and supported by Ministry of Education (MOE) and with collaboration of the Japan Society for the Promotion of Science (JSPS) Asian CORE programme and the Establishment of Research Education Network on Coastal Marine Science in Southeast Asia (COMSEA).
REFERENCES CITED
Agu, C. V., Njoku, O. V., Chilaka, F. C., Okorie, S. A., and Agbiogwu, D. (2012). “Physico-chemical characterization of lignocellulosic fibre from Ampelocissus cavicaulis,” Int. J. Basic & Appl. Sci. IJBAS-IJENS 12(3), 68-77.
Akgul, M., and Tozluoglu, A. (2009). “Some chemical and morphological properties of juvenile woods from beech (Fagus orientalis L.) and pine (Pinus nigra A.) plantations,” Trends in Applied Sciences Research 4(2), 116. DOI: 10.3923/TASR.2009.116.125
Akpakpan, A. E., Akpabio, U. D., and Obot, I. B. (2012). “Evaluation of physicochemical properties and soda pulping of Nypa fruticans frond and petiole,” Elixir Appl. Chem. 45, 7664-7668.
Alava, M., and Niskanen, K. (2006). “The physics of paper,” Reports on Progress in Physics 69(3), 669. DOI: 10.1088/0034-4885/69/3/R03
Bakker, R. R., and Elbersen, H. W. (2005). “Managing ash content and quality in herbaceous biomass: An analysis from plant to product,” 14th European Biomass Conference Paris, France, p. 17-21.
Barrett, T. (1983). Japanese Papermaking: Traditions. Tools, and Techniques, Weatherhill publisher, New York and Tokyo, p. 328.
Baydoun, E. A. H., and Brett, C. T. (1985). “Comparison of cell wall compositions of the rhizomes of three seagrasses,” Aquatic Botany, 23(2), 191-196. DOI: 10.1016/0304-3770(85)90065-8
Bektas, İ., and Tutus, A. (1999). “A study of the suitability of Calabrian pine (Pinus brutia Ten.) for pulp and paper manufacture,” Turkish Journal of Agriculture and Forestry 23(EK3), 589-598.
Biermann, C. J. (1996). Handbook of Pulping and Papermaking, Academic Press, United Kingdom, p. 754.
Bjorndal, K. A. (1980). “Nutrition and grazing behavior of the green turtle Chelonia mydas,” Marine Biology 56(2), 147-154. DOI: 10.1007/BF00397131
Cordeiro, N., Belgacem, M. N., Torres, I. C., and Moura, J. (2004). “Chemical composition and pulping of banana pseudo-stems,” Industrial Crops and Products 19(2), 147-154. DOI: 10.1016/J.INDCROP.2003.09.001
Cunha, A. H., Assis, J. F., and Serrao, E. A (2013). “Seagrass in Portugal: A most endangered marine habitat,” Aquatic Botany 104(2013), 193-203. DOI: 10.1016/j.aquabot.2011.08.007
Cunning, S. (1989). Handmade Paper: A Practical Guide to Oriental and Western Techniques, Cunning Enterprises, Raven’s Word Press, p. 113.
Davies, P., Morvan, C., Sire, O., and Baley, C. (2007). “Structure and properties of fibres from sea-grass (Zostera marina),” Journal of Materials Science 42(13), 4850-4857. DOI: 10.1007/S10853-006-0546-1
Eroglu, H. (1980). “Investigating possibilities of obtaining wood pulp from wheat straw by O2-NaOH method,” Ph.D Thesis, Karadeniz. Technical University.
Falanruw, M. C. (1992). “Seagrass nets,” Atoll Research Bulletin (USA), 364, 1-12.
Hatakka, A., Mettälä, A., Härkönen, T., and Paavilainen, L. (1996). “Biopulping of gramineous plants by white-rot fungi,” International Conference; 6th, Biotechnology in the Pulp and Paper Industry, Vienna, Austria, p. 229-232.
Hemmasi, A. H., Samariha, A., Tabei, A., Nemati, M., and Khakifirooz, A. (2011). “Study of morphological and chemical composition of fibers from Iranian sugarcane bagasse,” American-Eurasian J. Agric. & Environ. Sci., 11(4), 478-481.
Hunsigi, G. (1989). “Agricultural fibres for paper pulp,” Outlook on Agriculture 18, 96-103.
Hurter, A. M. (1990). “Utilisation of annual plants and agricultural residues for the production of pulp and paper,” Nonwood Plant Fibers Pulping Progress Report No. 19, TAPPI Press, Atlanta, Georgia, USA.
IAWA (1937). “Standard terms of length of vessel members and wood fibers,” Tropical Woods, 51, 21-22.
Jahan, M. S., and Rawshan, S. (2009). “Reinforcing potential of Jute pulp with Trema orientalis (Nalita) pulp,” BioResources 4(3), 921-931.
Janson, J., Jousimaa, T., Hupa, M., and Backman, R. (1996). “Phosphate-based pulping of agrofibre including papermaking and spent liquor recovery,” Nordic Pulp and Paper Research Journal 11(1), 005-009. DOI: 10.3183/NPPRJ-1996-11-01-p004-009
Japar Sidik, B., and Muta Harah, Z. (2003). “The seagrass of Malaysia,” in: World Atlas of Seagrasses, E. P. Green and F. T. Short (eds.), California University Press, California, pp. 152-160.
Jiménez, L., López, F., and Martínez, C. (1993). “Paper from sorghum stalks,” Holzforschung-International Journal of the Biology, Chemistry, Physics and Technology of Wood 47(6), 529-533. DOI: 10.1515/HFSG.1993.47.6.529
Jiménez, L., Rodríguez, A., Pérez, A., Moral, A., and Serrano, L. (2008). “Alternative raw materials and pulping process using clean technologies,” Industrial Crops and Products 28(1), 11-16. DOI: 10.1016/J.INDCROP.2007.12.005
Kang, T., and Paulapuro, H. (2006). “Effect of external fibrillation on paper strength,” Pulp and Paper Canada-Ontario 107(7/8), 51.
Kaur, H., and Dutt, D. (2013). “Anatomical, morphological and chemical characterization of lignocellulose by-products of lemon and sofia grasses obtained after recuperation of essential oils by steam distillation,” Cellulose Chemistry and Technology 47(1-2), 83-94.
Khiari, R., Mhenni, M. F., Belgacem, M. N., and Mauret, E. (2010). “Chemical composition and pulping of date palm rachis and Posidonia oceanica–A comparison with other wood and non-wood fibre sources,” Bioresource Technology 101(2), 775-780. DOI: 10.1016/J.BIORTECH.2009.08.079
Knoshaug, E. P., Sestric, R., Jarvis, E., Chou, Y.-C., Pienkos, P. T., and Darzins, A. (2009). “Current status of the department of energy’s aquatic species program lipid-focused algae collection,” Conference poster presentation to the 31st Symposium on Biotechnology for Fuels and Chemicals, National Renewable Energy Laboratory, May 2009.
Knoshaug, E. P., Shi, B., Shannon, T. G., Mleziva, M. M., and Pienkos, P. T. (2013). “The potential of photosynthetic aquatic species as sources of useful cellulose fibers—A review,” Journal of Applied Phycology 25(4), 1123-1134. DOI: 10.1007/S10811-012-9958-2
Kuo, J., and den Hartog, C. (2001). “Seagrass taxonomy and identification key,” in: Global Seagrass Research Methods, F. T. Short, R. G. and Coles (eds.), Elsevier, Amsterdam, Netherlands, pp. 31-58. DOI: 10.1016/B978-044450891-1/50003-7
Leopold, B., and Marton, R. (1974). Papermaking Potential of Zostera and Cladophora, Two Marine Weeds, New York Sea Grant Institute, Albany, New York, p. 12.
Madsen, B., Lilholt, H., Damkilde, L., Hoffmeyer, P., and Thomsen, A. B. (2004). Properties of Plant Fiber Yarn Polymer Composites: An Experimental Study, Report no. 082. Department of Civil Engineering, Technical University of Denmark.
Manzanares, M., Tenorio, J. L., and Ayerbe, L. (1997). “Sowing time, cultivar, plant population and application of N fertilizer on kenaf in Spain’s central plateau,” Biomass and Bioenergy 12(4), 263-271. DOI: 10.1016/S0961-9534(96)00078-5
Marques, G., Rencoret, J., Gutiérrez Suárez, A., and Río Andrade, J. C. D. (2010). “Evaluation of the chemical composition of different non-woody plant fibers used for pulp and paper manufacturing,” The Open Agriculture Journal 3, 1-9. DOI: 10.2174/1874331501004010093
Milchakova, N. A., Böer, B., Boyko, L. I., and Mikulich, D. V. (2014). “The chemical composition and technological properties of seagrasses a basis for their use (A review),” in: Sabkha Ecosystems: Volume IV: Cash Crop Halophyte and Biodiversity Conservation, M. Ajmal Khan, B. Boer, M. Ozturk, T. A. Al Abdessalaam, M. Clusener-Godt, and B. Gul (eds.), pp. 313-323. Springer, New York, London. DOI: 10.1007/978-94-007-7411-7_22
Moubasher, M. H., Abdel-Hafez, S. H., and Mohanram, A. M. (1982). “Direct estimation of cellulose, hemicellulose, lignin,” Journal of Agricultural Research 46, 1467-1476.
Nazhad, M. M., Harris, E. J., Dodson, C. T., and Kerekes, R. J. (2000). “The influence of formation on tensile strength of papers made from mechanical pulps,” Tappi Journal 83(12), 1-9.
Ogunwusi, A. A. (2014). “Agricultural waste pulping in Nigeria: Prospects and challenges,” Civil and Environmental Research 6(10), 101-110.
Ohshima, J., Yokota, S., Yoshizawa, N., and Ona, T. (2005). “Examination of within-tree variations and the heights representing whole-tree values of derived wood properties for quasi-non-destructive breeding of Eucalyptus camaldulensis and Eucalyptus globulus as quality pulpwood,” Journal of Wood Science 51(2), 102-111. DOI: 10.1007/s10086-004-0625-3
Oinonen, H., and Koskivirta, M. (1999). “Special challenges of pulp and paper industry in Asian populated countries, like Indian sub-continent and China,” Proceedings of the Paperex, pp. 49-68.
Ona, T., Sonoda, T., Ito, K., Shibata, M., Tamai, Y., Kojima, Y., Ohshima, J., Yokota, S., and Yoshizawa, N. (2001). “Investigation of relationships between cell and pulp properties in Eucalyptus by examination of within-tree property variations,” Wood Science and Technology 35(3), 229-243. DOI: 10.1007/s002260100090
Paavilainen, L., and Torgilson, R (1994). “Reed canary grass – A new Nordic papermaking fibre,” Proceedings of the TAPPI Pulping Conference, San Diego, California, USA, p. 611–618.
Saijonkari-Pahkala, K. (2008). “Non-wood plants as raw material for pulp and paper,” Agricultural and Food Science in Finland 10, 11-101.
Seo, Y. B., Lee, Y. W., Lee, C. H., and You, H. C. (2010). “Red algae and their use in papermaking,” Bioresource Technology 101(7), 2549-2553. DOI: 10.1016/J.BIORTECH.2009.11.088
Shakhes, J., Zeinaly, F., Marandi, M. A. B., and Saghafi, T. (2011). “The effects of processing variables on the soda and soda-AQ pulping of kenaf bast fiber,” BioResources 6(4), 4626-4639. DOI: 10.5897/AJBR11.023
Sharma, M., Sharma, C. L., and Kumar, Y. B. (2013). “Evaluation of fiber characteristics in some weed of Arunachal Pradesh, India for pulp and paper making,” Res. J. Agriculture and Forestry Sci. 1(3), 15-21.
Shatalov, A. A., Quilho, T., and Pereira, H. (2001). “Arundo donax L. reed: New perspectives for pulping and bleaching: I. Raw material characterization,” Tappi Journal 84(1), 45-61.
Shen, J., Zhou, X., Wu, W., and Ma, Y. (2012). “Improving paper strength by gelation of native starch and borax in the presence of fibers,” BioResources 7(4), 5542-5551. DOI: 10.15376/biores.7.4.5542-5551.
Silva, J. F., Duck, R. W., and Catarino, J. B. (2004). “Seagrass and sediment response to changing physical forcing in a coastal lagoon,” Hydrology and Earth System Science 8(2004), 151-159. DOI: 10.5194/hess-8-151-2004.
Sirvio, J., and Nurminen, I. (2004). “Systematic changes in paper properties caused by fines,” Pulp and Paper Canada-Ontario 105, 39-42.
Sood, Y. V., Pande, P. C., Tyagi, S., Payra, I., and Kulkarni, N. A. G. (2005). “Quality improvement of paper from bamboo and hardwood furnish through fiber fractionation,” Journal of Scientific & Industrial Research 64(4), 299-305.
Spiegelberg, H. L. (1966). “The effect of hemicelluloses on the mechanical properties of individual pulp fibers,” Tappi 49(9), 388-396.
Torbatinejad, N., and Sabin, J. R. (2010). “Laboratory evaluation of some marine plants on South Australian beaches,” Journal of Agricultural Science and Technology 3, 91-100.
Touzinski, G. F., Clark, T. F., Tallant, W. H., and Knolek, W. F. (1973). “Soda pulps from kenaf bark and from core,” CA report no. 52, TAPPI non-wood plant fibre pulping progress report, Atlanta, Georgia.
Tutuş, A., Kazaskeroğlu, Y., and Çiçekler, M. (2015). “Evaluation of tea wastes in usage pulp and paper production,” BioResources 10(3), 5407-5416. DOI: 10.15376/biores.10.3.3818-3830
Ververis, C., Georghiou, K., Christodoulakis, N., Santas, P., and Santas, R. (2004). “Fiber dimensions, lignin and cellulose content of various plant materials and their suitability for paper production,” Industrial Crops and Products 19(3), 245-254. DOI: 10.1016/J.INDCROP.2003.10.006
Wahab, R., Mustafa, M. T., Rahman, S., Salam, M. A., Sulaiman, O., Sudin, M., and Rasat, M. S. M. (2006). “Relationship between physical, anatomical and strength properties of 3-year-old cultivated tropical bamboo Gigantochloa scortechinii,” Journal of Agricultural and Biological Science 7(10), 782-791.
Wathen, R. (2006). “Studies on fiber strength and its effect on paper properties,” KCL Communications 11, 1-97. KL, Espoo, Finland.
Wood, I. M. (1981). “The utilization of field crops and crop residues for paper pulp production,” Field Crop Abstract 34, 557-568.
Xu, F., Zhong, X. C., Sun, R. C., and Lu, Q. (2006). “Anatomy, ultrastructure and lignin distribution in cell wall of Caragana korshinskii,” Industrial Crops and Products 24(2), 186-193. DOI: 10.1016/j.indcrop.2006.04.002
Young, J. H. (1981). “Fiber preparation and approach flow in pulp and paper,” in: Chemistry and Chemical Technology, Interscience Publishers, New York, pp. 25-35.
Article submitted: October 7, 2015; Peer review completed: January 22, 2016; Revised version received and accepted: April 14, 2016; Published: May 2, 2016.
DOI: 10.15376/biores.11.2.5358-5380
