Abstract
A multifunctional fluorescent polymer based on coumarin (MFPC) was synthesized using a four-step synthetic route. To obtain the target compound, 7-amino-4-methylcoumarin, coumarin fluorescent monomer (CFM), and ultraviolet absorber monomer (UVAM) were prepared. By the reaction of CFM, UVAM, trimethyl-2-methacroyloxyethylammonium chloride (DMC), and acrylamide (AM) as a linker, a new multifunctional fluorescent polymer was synthesized. The structure of the MFPC was characterized by Fourier transform infrared (FTIR) and nuclear magnetic resonance (1H-NMR) spectroscopies. The optical properties of MFPC were measured by ultraviolet-visible (UV-vis) and fluorescence spectroscopies, in aqueous solution. The integration effect between MFPC and paper was tested by scanning electron microscopy. The application performance of MFPC on paper was evaluated by measuring the surface strength, smoothness, and whiteness using the printability tester and the UV accelerated aging test. Results indicated that the MFPC had good solubility in water. It also exhibited a positive effect on the light stability and surface strength of paper as a light stabilizer, fluorescent brightener, and surface sizing agent.
Download PDF
Full Article
Synthesis and Application of a Multifunctional Fluorescent Polymer Based on Coumarin
Guanghua Zhang,a,b,* Hua Zheng,a,b Guojun Liu,a,b Peng Wang,a,b and Rui Xiang a,b
A multifunctional fluorescent polymer based on coumarin (MFPC) was synthesized using a four-step synthetic route. To obtain the target compound, 7-amino-4-methylcoumarin, coumarin fluorescent monomer (CFM), and ultraviolet absorber monomer (UVAM) were prepared. By the reaction of CFM, UVAM, trimethyl-2-methacroyloxyethylammonium chloride (DMC), and acrylamide (AM) as a linker, a new multifunctional fluorescent polymer was synthesized. The structure of the MFPC was characterized by Fourier transform infrared (FTIR) and nuclear magnetic resonance (1H-NMR) spectroscopies. The optical properties of MFPC were measured by ultraviolet-visible (UV-vis) and fluorescence spectroscopies, in aqueous solution. The integration effect between MFPC and paper was tested by scanning electron microscopy. The application performance of MFPC on paper was evaluated by measuring the surface strength, smoothness, and whiteness using the printability tester and the UV accelerated aging test. Results indicated that the MFPC had good solubility in water. It also exhibited a positive effect on the light stability and surface strength of paper as a light stabilizer, fluorescent brightener, and surface sizing agent.
Keywords: 7-Amino-4-methyl coumarin; Multifunctional fluorescent polymer; Light stability; Surface strength; High-yield pulp
Contact information: a: Key Laboratory of Additives of Chemistry & Technology for Chemical Industry, Ministry of Education, Shaanxi University of Science & Technology, Xi’an 710021, Shaanxi, China; b: College of Chemistry & Chemical Engineering, Shaanxi University of Science & Technology, Xi’an 710021, Shaanxi, China; *Corresponding author: zhanggh@sust.edu.cn
INTRODUCTION
In recent years, high-yield pulp (HYP) has received increasing interest as a substitute for bleached hardwood kraft pulp in the manufacturing of uncoated fine paper, coated fine paper, and paperboards. This is because of the unique properties of HYP viz., high bulk, high opacity, high stiffness, and good printability. However, HYP contains much more lignin and hemicelluloses, which could have a negative effect on the light stability of paper made from HYP under certain external environments, such as thermal induction, light induction, and the presence of metal cations (Cockram 1989; Cannell and Cockram 2000; Ni et al. 2011; Manda et al. 2012). Because of the light-induced yellowing mechanism of HYP, many methods have been studied all over the world, such as chemical modification of lignin, exploration of new bleaching processes of high yield pulp, and the addition of chemical auxiliaries. From the perspective of cost and the effect of light stability, adding chemical auxiliaries (ultraviolet absorbents, radical scavengers, fluorescent whitening agents, etc.) has become the most feasible method to inhibit the yellowing of HYP (Bourgoing et al. 2001; Zhang et al. 2009; Fatehi et al. 2010; Zhang et al. 2011b; Shi et al. 2012; Chen et al. 2013).
In our laboratory, multiple studies have been carried out to inhibit the yellowing of HYP (Liu et al. 2011a,b; Guo et al. 2012; Zhang et al. 2012; Wang et al. 2013b, 2014a; Liu et al. 2015). Although these works have made some achievements in the field of paper yellowing inhibition, there are still some shortcomings, such as bad solubility in water and poor retention on the surface of paper. Besides, they require starch adhesive when coated on the surface of paper. In order to overcome these deficiencies, many methods have been adopted. For instance, the introduction of water-soluble compounds or polymerization (Bangar and Varandajaran 1994; Wagner 2009; Guo et al. 2012; Zhang et al. 2011a; Cigan et al. 2013; Essaidi et al. 2013). In this paper, a novel multifunctional fluorescent polymer based on coumarin (MFPC) was synthesized by copolymerization, containing CFM, UVAM, DMC, and AM monomers. As a cationic monomer, DMC can provide improvements in adhesion on the surface of paper on account of its high polarity and AM plays an important role in the copolymerization as a bridge (Linhart et al. 2005; Cao et al. 2006; Wang et al. 2013a,b, 2014a), which make the water-insoluble coumarin fluorescent monomer turn into a multifunctional polymer with water-soluble and adhesive properties. Also, its multifunctional properties on paper were evaluated as a light stabilizer, fluorescent brightener, and surface sizing agent.
EXPERIMENTAL
Materials
The main raw materials used to synthesize the 7-amino-4-methylcoumarin, CFM, and UVAM were as follows: 3-aminophenol (Sinopharm Chemical Reagent Co. Ltd, Shang hai), ethyl acetoacetate, zinc chloride (ZnCl2), anhydrous ethanol (CH3CH2OH), concentrated sulfuric acid (H2SO4), isopropanol ((CH3)2CHOH), chloroform (CHCl3), triethylamine,2,4-dihydroxybenzophenone (UV-0), acetone, sodium bicarbonate (NaHCO3), acryloyl chloride, ammonium persulfate ((NH4)2S2O8), urea (CO(NH2)2), trimethyl-2-methacroyloxyethylammonium chloride (DMC), acrylamide (AM) (analytical grade, Tianli Chemical Reagent Co. Ltd, Tian jin), and poplar CMP (Yueyang Paper Ltd., China).

Fig. 1. Schematic diagram of synthetic route of 7-amino-4-methylcoumarin, CFM, and UVAM
Methods
Synthesis of 7-amino-4-methylcoumarin, CFM, and UVAM
Based on the Pechmann method (Lanman 2011), 7-amino-4-methyl coumarin was synthesized by the reaction of 3-aminophenol (9.7 g) and ethyl acetoacetate (5.45 g) in the presence of zinc chloride (8.5 g) as a catalyst and anhydrous ethanol (30 mL) as a solvent. Through the reaction of 7-amino-4-methylcoumarin (5 g), acryloyl chloride (3.1 g), triethylamine (8.7 g) as a base, and trichloromethane (50 mL) as a solvent, the CFM was obtained. In the same way, by using 2,4-dihydroxybenzophenone (5.35 g), acryloyl chloride (3.62 g), triethylamine (2.6 g) as a base, and acetone (30 mL) as a solvent, the CFM was synthesized.
Synthesis of MFPC
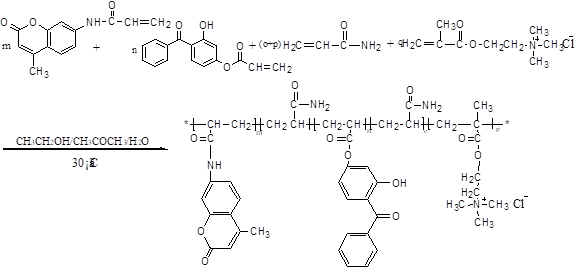
Fig. 2. Schematic diagram of synthetic route of MFPC (m, n, o, p, q = k-1, k=0, 1, 2, 3 ……)
The synthetic route and molecular structure of MFPC are shown in Fig. 2. Under the (NH4)2S2O8 -CO(NH2)2 oxidation-reduction system (W (NH4)2S2O8):W CO(NH2)2 =1:1), CFM, UVAM, DMC, and AM (the molar ratio: n CFM: n UVAM: n DMC: n AM = 0.5:0.5: 5: 4 ) were combined with a mixture of ethanol-water solution as solvent in a 250-mL round-bottom flask equipped with a stirring bar and a thermometer under nitrogen protection. The mixture was stirred for 6 h while maintaining the temperature at 30 °C. At the end of the reaction, MFPC was isolated by the evaporation of the solvents in a rotary evaporator under vacuum. Then, the product was recrystallized with trichloromethane and acetone to remove the unreacted monomers and dried under vacuum overnight at 80 °C.
1HNMR: (400MHz, DMSO, 40 °C): d/ppm: 6.90 to 7.57 (H of benzene), 5.58 (NH2 of AM), 4.06 (OH of UV), 2.48 (CH3 of coumarin), 2.92 to 3.40 (CH3 of DMC), 1.24 to 2.09 (CH2 and CH3 of the main chain). I.R. (KBr): 3419 cm-1 (NH stretch); 2958 cm-1 (CH2 stretch); 1727 cm-1(C=O of coumarin stretch); 1668 cm-1 (AM stretch); 1529 cm-1, 1479 cm-1, and 1417 cm-1 (Ar stretch); 1482 cm-1 (CH2-N(CH3)3 of the DMC deformation); 1269 cm-1 and 1222 cm-1 (C-O-C stretch).
Preparation of paper sheets
Using deionized water, pulp was prepared to 10% concentration (based on dry pulp); then, 1% H2O2, 0.05% EDTA, and 0.5% Na2SiO3.9H2O (based on dry pulp) were added to adjust the pH to the range of 9 to 10. The pulp was kept in hot water (70 °C) for 90 min; it was then washed with deionized water to neutral pH. Paper sheets (100 g·m-2) were made with the pulp and cut into pieces (68 mm × 73 mm). Surface sizing material was prepared by adding the synthesized compounds into the solution. The paper sheets were coated on the coating machine (ST-1-260, Shaanxi University of Science & Technology, China) with the sizing materials. In the opaque background, the paper samples were dried at room temperature.
The Stokes shift
The Stokes shift is a parameter that indicates the difference in the properties and structure of the fluorophores between the ground state S0 and the first excited state S1. A larger Stokes shift indicates higher energy losses in the electron transition process. The Stokes shifts (cm-1) were calculated using Eq. 1.
![]()
Characterization
Infrared spectra analyses were recorded on a Vector-22 FT-IR spectrometer (Bruker Corporation, Germany) with KBr pellets. Proton-NMR spectra were measured on a Bruker Avance III HD 400 MHz NMR spectrometer (Bruker Corporation, Germany) in DMSO solution with tetramethylsilane (TMS) as the internal standard. Ultra-violet spectra were measured using a Cary 100 UV-Visible Spectrophotometer (Agilent, USA). Fluorescence spectra were measured on a Fluorolog spectrofluorometer (Horiba, France). Zeta potential was recorded using a Zetasizer NANO-ZS90 (Malvern, UK). The morphology of paper sheets was observed using S4800 SEM images (Rigaku, Japan).
Surface sizing experiment
Deionized water was added to the MFPC to prepare solutions with concentrations of 0.2%, 0.4%, 0.6%, and 0.8%. The papers were coated with the solutions mentioned above in a coating machine and dried in a glazing machine. The surface strength of coated paper was measured by a J-IGT350 printability tester (Sichuan Changjiang Instrument Co. Ltd, China). According to the national standard method (Liu 2004), all the quality indicators of the patterns were measured.
UV accelerated aging test
The whiteness of the paper sheets was determined by a ZB-A colorimeter (Hangzhou Zhibang Instrument Co. Ltd, China) at R457 nm. The whiteness numerical value was obtained as the average value of five runs.
Paper anti-UV aging testing was carried out using a ZN-100N UV (Xi’an Tongsheng Instrument Manufacturing Co. Ltd., China) light resistant climate chamber (tube of UV-340). The test conditions used were as follows: 25 °C, wavelength of UV lamp 340 nm, output power 5.3 mW·cm-2, radiation for 48 h, and distance between the samples and lamp 30 cm. The whiteness of the samples was determined at set intervals.
The post color number (PC) is usually used to represent the level of anti-UV aging of paper, which can reflect the relative quantities of the chromogenic substances during the process of the UV accelerated aging test. The PC value was calculated using the following Eqs. 2 and 3 (Zhang 2008):

where k and s denote the light absorption and scattering coefficients, respectively; R∞ represents the whiteness measured in R457; and the subscripts o and t denote the initial and final UV aging, respectively.
RESULTS AND DISCUSSION
UV-vis Spectra
The UV-vis spectra of CFM, UVAM, and MFPC in water solution at a concentration of 2.0×10-4 kg·L-1 are shown in Fig. 3; the UV-vis region is from 200 to 800 nm. MFPC has two peaks (peak1 and peak 2), and the maximum absorption wavelengths of peak1 and peak2 are 289 and 324 nm, respectively. As is illustrated in Fig. 3, the absorbance of MFPC is better than those of CFM and UVAM. At the same dosage of CFM, UVAM, and MFPC, the solutions of CMF and UVAM reach the saturation state. However MFPC can be completely dissolved in water, forming a dilute solution. So MFPC solution obeys Beer’s law and could absorb more ultraviolet light. In addition, MFPC also possesses a wider ultraviolet absorption range because MFPC contains the groups of CFM and UVAM.
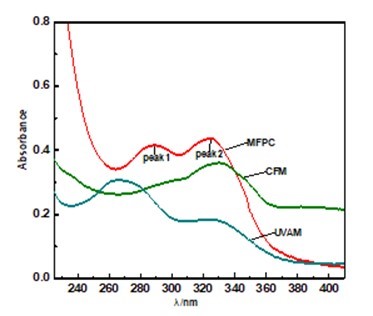
Fig. 3. The UV-vis spectra of CFM, UVAM, and MFPC
Although DMC, as a quaternary ammonium salt, could decrease the conjugated system and π-electron delocalization to a certain extent, the copolymerization of DMC/AM/CFM/UVAM increases the conjugate groups of the main chain of the polymer, which broadens the π-electron delocalization and reduces the π-π* transition band width. As is well known, 290 to 320 nm in the ultraviolet region is most likely to cause the photo-yellowing of paper. The MFPC solution would be able to absorb this part of ultraviolet light, which can effectively inhibit the color reversion of paper.
To reveal the good dispersion of MFBC in water, the absorbance at 324 nm against the concentration of MFPC is plotted in Fig. 4. The absorbance at 324 nm is plotted against the concentration of MFPC. Obviously, the MFPC solution obeys Beer’s law, and the extinction coefficient is calculated to be 0.0598 mg-1cm-1. The linear relationship between concentration and absorption is an indication of the excellent dispersion of MFPC in water (Tang et al. 2011).
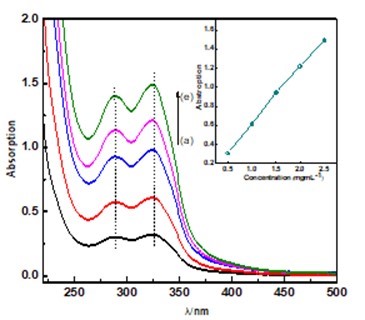
Fig. 4. UV-vis spectra of MFPC dispersed in water at various concentrations (concentrations are 0.5, 1.0, 1.5, 2, 2.5 mgmL-1 from a to e, respectively). The inset shows the linear relationship between absorption at 324 nm and the concentration of MFPC.
Fluorescence Spectra
The absorption and fluorescence spectra of CFM and MFPC are shown in Fig. 5. The absorption (λA) and fluorescence (λF) maxima of CFM and MFPC are, respectively, 334 nm and 394 nm, and 338 nm and 393 nm.
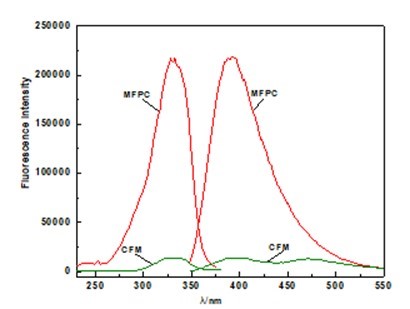
Fig. 5. The absorption and fluorescence spectra of CFM and MFPC (c =2.0×10-4 Kg·L-1)
The Stokes shift is an important characteristic of fluorescent compounds. As is shown in Fig. 5, the value for the Stokes shift of CFM (Δσ = 4559.41 cm-1) is much higher than the value obtained for MFPC (Δσ = 4010.35 cm-1), which indicates that the structure of MFPC is more stable and the light stability is better than that of CFM (Magalhaes et al. 2006; Donovalova et al. 2012).
The ζ Potential of MFPC
The ζ potential is an important index denoting the cationic degree of a reaction liquid. With increasing concentration of MFPC, the ζ potential of samples becomes larger, which indicates that the cationic degree of MFPC becomes larger, as shown in Fig. 6. It is well-known that paper fibers typically have negative charge. When MFPC with a positive charge is coated on the surface of paper, an electrostatic attraction between MFPC and paper fibers will be generated. Therefore, MFPC could be easy to retain on the surface of the paper, which would enhance the surface strength of paper and improve the anti-UV aging effect.
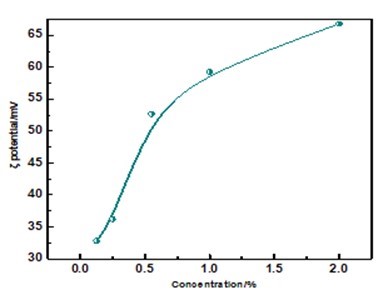
Fig. 6. The ζ potential of MFPC (concentrations are 0.125, 0.25, 0.5, 1, 2 g/L from C1 to C5, respectively)
SEM Images of Surface Sized Paper
SEM images (Fig. 7) demonstrate a representative overview of blank paper sheets (Fig. 7a) and paper sheets with MFPC (Fig. 7b). The fibers of blank paper sheets combine more loosely, leading to a lower smoothness of the paper. After adding MFPC on the paper sheets, the fibers of the paper sheets with MFPC combine more closely, and the smoothness of coated paper becomes better than that of blank paper sheets. The combination points of the fiber are also increased, which improves the tightness and surface strength of the paper sheets.

Fig. 7. SEM images of surfaces of blank paper sheet and surface sized paper
Surface Sizing Experiment
The surface strength and smoothness are important indicators for print paper. As shown in Figs. 8 and 9, papers coated with various concentrations of MFPC exhibited higher surface strength and smoothness than uncoated paper. Compared with uncoated paper, whose surface strength and smoothness are 0.79 m/s and 37.6 s, respectively, the surface strength of coated paper with 0.2%, 0.4%, 0.6%, and 0.8% concentrations of MFPC, respectively, increased by 0.73, 1.11, 1.45, and 1.76 m/s.
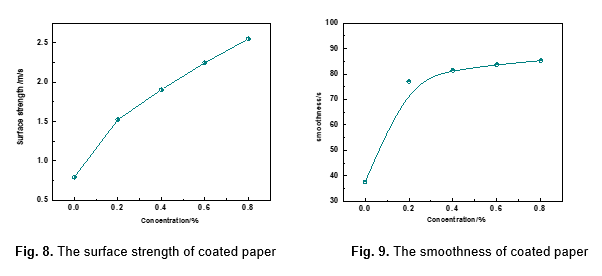
Meanwhile, the smoothness of coated paper, respectively, increased by 39.5, 43.9, 46.1, and 47.7 s. Therefore, with an increase in the concentration of MFPC, the surface strength and smoothness of coated paper were intensified, which indicates that MFPC has good film-forming property and can be applied in the paper making industry as a surface sizing agent (Wang et al. 2014b, 2015). (The coating weights of MFPC were 0.1, 0.2, 0.3, and 0.4 g/ m2, while the dosages of the MFPC were 0.2%, 0.4%, 0.6%, and 0.8%).
UV Accelerated Aging Test
Paper sheets coated by 0.8% CFM, UVAM, MFPC, and PFCC (Liu et al. 2015) were investigated in the contrast test.
During the 48-h UV accelerated aging test, the brightness and the PC value of paper sheets were measured, and the results are shown in Figs. 10 and 11. (The coating weight of CFM, UVAM, MFPC, and PFCC was 0.4 g/ m2, while the dosage of the products was 0.8%.)
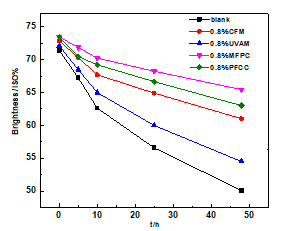
Fig. 10. The change of brightness of paper sheets with UV irradiation time
After 48 h of UV aging, the brightness of blank paper sheet and paper sheets coated with CFM, UVAM, MFPC, and PFCC were, respectively, 50.09, 61.02, 54.51, 65.46, and 63.03 ISO%. The brightness of paper sheets, respectively, decreased by 21.32, 11.84, 17.60, 8.03, and 10.46 ISO% compared with the initial brightness.
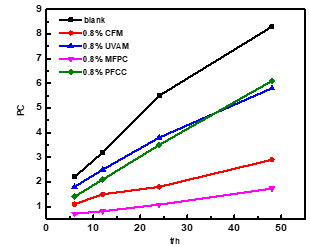
Fig. 11. The change in PC value of paper sheets with UV irradiation time
In addition, the PC values of the blank paper sheet and the coated sheets were respectively 8.3, 2.9, 5.8, 1.7, and 6.1. It is well known that the UV-0 structure can absorb UV radiation, and the coumarin structure not only absorbs UV radiation, but also increases the initial brightness. Therefore, the CFM and UVAM have anti-UV aging properties. Compared with CFM and UVAM, MFPC showed a greater anti-UV aging effect under the synergistic effect of CFM and UVAM, exhibiting excellent application performance for the paper making industry as a light stabilizer and fluorescent brightener. MFPC also showed a greater anti-UV aging effect than PFCC. The difference in anti-UV aging effects between MFPC and PFCC may result from a difference in the water solubility and electric properties.
CONCLUSIONS
In conclusion, a novel multifunctional fluorescent polymer as light stabilizer, fluorescent brightener, and surface sizing agent was synthesized by a four-step synthetic route.
- According to the UV and fluorescence spectra, MFPC not only has good solubility in water, but also exhibits excellent luminescent properties. Its Stokes shift is less than that of CFM, which indicates that the multifunctional fluorescent polymer has little change in steric hindrance, loses less energy, and possesses a better photostability than
- The surface sizing experiment indicates that MFPC can be applied to enhance the surface strength and smoothness of paper as a surface sizing agent, which is due to its good film-forming properties.
- The UV aging experiment further demonstrates that MFPC has a better resistance to UV aging as a light stabilizer and fluorescent brightener, resulting from the synergistic effect of CFM and UVAM.
- Compared with the PFCC synthesized in our research laboratory, MFPC not only has better solubility in water, but also exhibits multifunctionality as a light stabilizer, fluorescent brightener, and surface sizing agent.
ACKNOWLEDGMENTS
This work was supported by the National Natural Science Foundation of China (21176148, 21303098), the Natural Science Foundation of Shaanxi Province of China (2009JM2010), and the R&D Center for Green Light Chemical Additives Foundation of Transfer Group of China (No. 20124932)
REFERENCES CITED
Bangar, R. B., and Varandajaran, T. S. (1994). “Substituent and solvent effects on the twisted intramolecular charge transfer of three new 7-(diethylamino) coumarin-3-aldehyde derivatives,” J. Phys. Chem. 98(36), 8903-8905. DOI: 10.1021/j100087a014
Bourgoing, S., Leclerc, E., and Martin, P. (2001). “Use of fluorescent whitening agents to inhibit light-induced colour reversion of unbleached mechanical pulps,” J. Pulp Pap. Sci. 27(7), 240-244.
Cannell, E., and Cockram, R. (2000). “The future of BCTMP,” Pulp and Paper 74(5), 61, 63, 65-66, 68-70, 72, 74-76.
Cao, C. B., Han, H. B., and Wang, D. Y. (2006). “Synthesis and properties of quaternary ammonium salt fluorescent brighteners,” Chin. J. Lumin (Chinese Edition) 57(12), 3010-3015.
Chen, J., Zhang, M. Y., and Yuan, Z. R. (2013). “Improved yield pulp network and paper sheet properties by the addition of fines,” BioResources 8(4), 6309-6322. DOI: 10.15376/biores.8.4.6309-6322
Cigan, M., Donovalova, J., and Szocs, V. (2013). “7-(Dimethylamino)coumarin-3-carbaldehyde and its phenylsemicarbazone: TICT excited state modulation, fluorescent H-aggregates, and preferential salvation,” J. Phys. Chem. A 117(23), 4870-4883. DOI: 10.1021/jp402627a
Cockram, R. A. (1989). “CTMP in fine papers,” International Mechanical Pulping Conference Proceedings, Helsinki, pp. 20-24.
Donovalova, J., Cigan, M., Stankovicova, H., Gaspar, J., Danko, M., Gaplovsky, A. and Hrdlovic, P. (2012). “Spectral properties of substituted coumarins in solution and polymer matrices,” Molecules 17(3), 3259-3276. DOI: 10.3390/molecules17033259
Essaidi, Z., Krupka, O., and Iliopoulos, K. (2013). “Synthesis and functionalization of coumarin-containing copolymers for second order optical nonlinearities,” Opt. Mat. 35(3), 576-581. DOI: 10.1016/j.optmat.2012.10.011
Fatehi, P., Liu, X., and Ni, Y. H. (2010). “Interaction of cationic modified poly vinyl alcohol with high yield pulp,” Cellulose 17(5), 1021-1031. DOI: 10.1007/s10570-010-9432-8
Guo, J. G., Zhang, G. H., and Xu, H. L., (2012). “Synthesis and application of polymeric yellowing inhibitor based on naphthalimid fluorescent brightening agent,” J. Funct. Mater. (Chinese Edition) 43(23), 2881-2885.
Lanman, B. A. (2011). “Pechmann coumarin synthesis,” Name Reactions in Heterocyclic Chemistry II (2011), 454-469.
Linhart, F., Blum, T., and Dirks, B. (2005). “Activating fluorescent whitening agents with cationic polymers,” Professional Papermaking 2, 26-31.
Liu, G. J., Zhang, G. H., and Wang, P. (2015). “Synthesis and application of polymeric fluorescent compounds based on coumarin,” BioResources 10(3), 4784-4794. DOI: 10.15376/biores.10.3.4784-4794
Liu, J., and Zhang, G. H. (2011a). “Synthesis and photoproperties of fluorescent brighteners modified by polyaminamide,” Advanced Materials Research 233-235, 3064-3067.
Liu, J., and Zhang, G. H. (2011b). “The synthesis and study on optical characteristics of the asymmetrical polymerizable fluorescent brighteners,” International Journal of Polymeric Materials 60, 199-211. DOI: 10.1080/00914037.2010.504170
Liu, S. C. (2004). The Analysis and Detection of Pulp and Paper, Chemical Industry Press, Beijing.
Magalhaes, J. L., Pereira, R. V., and Triboni, E. R. (2006). “Solvent effect on the photophysical properties of 4-phenoxy-N-methyl-1,8-naphthalimide,” J. Photochem. Photobiol. A 183(1-2), 165-170. DOI: 10.1016/j.jphotochem.2006.03.012
Manda, B. M. K., Blok, K., and Patel, M. K. (2012). “Innovations in papermaking: An LCA of printing and writing paper from conventional and high yield pulp,” Sci. Total Environ. 439, 307-320. DOI: 10.1016/j.scitotenv.2012.09.022
Ni, Y. H., He, Z. B., and Zhang, H. J. (2011). “Characteristics of high yield pulp and its effect on some typical wet-end issue,” J. Biobased Mater. Bio. 5(2), 181-186. DOI: 10.1166/jbmb.2011.1134
Shi, H., Liu, H. B., and Ni, Y. H., (2012). “Review: Use of optical brightening agents (OBAs) in the production of paper containing high-yield pulps,” BioResources 7(2), 2582-2591. DOI: 10.15376/biores.7.2.2582-2591
Tang, Z. H., Zeng, C. F., Lei, Y. D., Guo, B. C., Zhang, L. Q., and Jia, D. M. (2011). “Fluorescent whitening agent stabilized graphene and its composites with chitosan,” J. Mater. Chem. 21, 17111-17118 .
Wagner, B. D. (2009). “The use of coumarins as environmentally sensitive fluorescent probes of heterogeneous inclusion systems,” Molecules 14(1), 210-237. DOI: 10.3390/molecules14010210
Wang, M. S., Cao, C. B., and Sun H. Y. (2013a). “Preparation of stibene-eighteen alkyl quaternary ammonium salts as fluorescent brightening materials and surfactants,” Adv. Mater. Res. 690-693,615-618.
Wang, P., Zhang, G. H., and Xiang, R., (2013b). “Preparation and optical properties of coumarin fluorescent polymer,” Chin. J. Lumin. 10, 1313-1318.
Wang, P., Zhang, G. H., and Xiang, R. (2014a). “Synthesis and application of polymer light stabilizer based on coumarin fluorescent brightener,” Polym. Mater. Sci. Eng. (Chinese Edition) 3, 25-29.
Wang, Z. S, Yu, W. L., and Dong, H. X. (2014b). “Study on solid surface sizing agent for paper,” Adv. Mater. Res. 884-885, 233-236.
Wang, H. H., Fan, J., and Fei G. Q. (2015). “Preparation and property of waterborne UV-curable chain-extended polyurethane surface sizing agent: Strengthening and waterproofing mechanism for cellulose fiber paper,” J. Appl. Polym. Sci. 132(31), 42354. DOI: 10.1002/app.42354
Zhang, R. (2008). “Synthesis of a novel yellowing inhibitor and study of its use for high-yield bamboo pulps,” Nanning: Guangxi University, (Chinese Edition).
Zhang, H. J., Hu, H. R., and Xu, Z. G. (2009). “Use of fluorescent whitening agents against light-induced color reversion of aspen BCTMP,” Appita J. 62(5), 355-359.
Zhang, Y. H., Zhang, Q. P., and Shi, W. Y. (2011a). “Synthesis and quantum chemistry study on fluorescence spectra of three 1,8-naphthalimide monomers,” Chin. J. Lumin. (Chinese Edition) 32(5), 505-513.
Zhang, Y., Ni, Y., and Wong, D. (2011b). “Distribution of option brightening agent (OBA), in the fiber wall of high-yield and kraft pulps,” J. Sci. Technol. Forest Prod. Process 1(1), 21-26.
Zhang, G. H., Xu, H. L., and Guo, J. G. (2012). “Synthesis and properties of new yellowing inhibitor with fluorescent whitening,” Huagong Xuebao (Chinese Edition) 63(10), 3358-3364. DOI: 10.3969/j.issn.0438-1157.2012.10.050
Article submitted: July 20, 2015; Peer review complete: September 19, 2015; Revised version received and accepted: October 31, 2015; Published: November 17, 2015.
DOI: 10.15376/biores.11.1.373-384
