Abstract
In the present work, enzymatic/mild acidolysis lignin (EMAL) was isolated from the raw material Cunninghamia lanceolata, and hydrothermal depolymerization was carried out in subcritical water (from 250 to 350 °C) using a cylindrical autoclave. The results revealed that the lowest yield of solid residue and highest liquid product yield were achieved at 325 °C. The liquid products were primarily composed of phenolic monomers and oligomers. As the reaction temperature increased, repolymerization of the liquid products and dehydrogenation and deoxidation of the solid residues occurred. The high heating value (HHV) of the residues was larger than that of the EMAL, and it reached a maximal level at 275 °C. Hydrothermal depolymerization and condensation reactions took place simultaneously even under mild conditions (250 °C). Carbonization of the EMAL was remarkable when the reaction temperature reached 325 °C.
Download PDF
Full Article
Temperature Impact on the Hydrothermal Depolymerization of Cunninghamia lanceolata Enzymatic/Mild Acidolysis Lignin in Subcritical Water
Yuan-Yuan Zhao, Xiao-Hong Li, Shu-Bin Wu,* and You-Ming Li
In the present work, enzymatic/mild acidolysis lignin (EMAL) was isolated from the raw material Cunninghamia lanceolata, and hydrothermal depolymerization was carried out in subcritical water (from 250 to 350 °C) using a cylindrical autoclave. The results revealed that the lowest yield of solid residue and highest liquid product yield were achieved at 325 °C. The liquid products were primarily composed of phenolic monomers and oligomers. As the reaction temperature increased, repolymerization of the liquid products and dehydrogenation and deoxidation of the solid residues occurred. The high heating value (HHV) of the residues was larger than that of the EMAL, and it reached a maximal level at 275 °C. Hydrothermal depolymerization and condensation reactions took place simultaneously even under mild conditions (250 °C). Carbonization of the EMAL was remarkable when the reaction temperature reached 325 °C.
Keywords: Subcritical water; Lignin; Depolymerization; Phenols; Biochar
Contact information: State Key Laboratory, Pulp & Paper Engineering, South China University of Technology Guangzhou, Guangdong, P. R. China 510640; *Corresponding author: shubinwu@scut.edu.cn
INTRODUCTION
In recent years, the high-value utilization of biomass is attracting attention, especially because of the ever-shrinking fossil fuel reserves and the enormous biomass stores worldwide. With a supply of approximately 200 billion metric tons per year worldwide, lignocellulose is the most abundant renewable biomass resource (García et al. 2009). Lignocellulosic biomass chemicals are promising as a substitute for petroleum chemicals in some industrial processes (Toledano et al. 2012). Lignin, one of the main components of lignocellulose (Toledano et al. 2010), is most abundant in aromatic compounds in nature and generates a large quantity of chemical reagents or adhesives, which can be applied as a replacement for chemicals refined from fossil oil (Toledano et al. 2012).
Recent attempts have been made to investigate the hydrothermal degradation of lignin model compounds (Kanetake et al. 2007; Okuda et al. 2008; Sasaki and Goto 2009, 2011) and lignin with a relatively low molecular weight (Fang et al. 2008; Sasaki and Goto 2008). Those works have been focused on clarifying the mechanism determining the reaction kinetics. It is believed that sub- and super-critical water has a positive effect on the degradation of lignin to phenols. To thoroughly investigate the hydrothermal depolymerization properties of lignin, it is necessary to isolate representative and unaltered lignin from biomass. At present, to isolate chemically unaltered lignin from wood, the most widely used techniques are based on the extraction of ball-milled wood by neutral solvents. Milled wood lignin (MWL) was extracted from finely milled wood without any previous treatment; cellulolytic enzyme lignin (CEL) was obtained by removing most of the carbohydrate with cellulolytic enzymes prior to aqueous dioxane extraction of ball-milled wood meal (Ikeda et al. 2002). On the basis of MWL and CEL, Wu and Argyropoulos (2003) put forward a modified method to separate lignin, i.e., combining enzymatic and mild acidolysis. This enzymatic mild acidolysis lignin (EMAL) is believed to be more representative, resulting in higher yield and purity (Wu and Argyropoulos 2003; Guerra et al. 2006). Meanwhile, the preparation procedure of EMAL is similar to the approach employed in cellulosic bio-refinery, and subcritical water treatment of EMAL is likely to play an important role in the bio-refinery industry.
In this study, the hydrothermal degradation reaction of Cunninghamia lanceolata EMAL was carried out within the temperature range of 250 to 350 °C for 30 min to explore its depolymerization characteristics. After the hydrothermal decomposition reaction, the liquid product (LP) and solid residual (SR) were separated, collected, and characterized. This research focused on the influence of temperature on EMAL and the character of the liquid fraction and residue during the reaction period.
EXPERIMENTAL
Lignin Preparation
Cunninghamia lanceolata was acquired from Guangdong Province (P. R. China). After removing the peel, Cunninghamia lanceolata was ground using a disintegrator and then extracted for 48 h with a benzene/ethanol solution (90:10 v/v). The wood powder was further milled in a porcelain jar for 10 days at a constant rotational speed of 36 rpm. The acquired fine powder was used to separate lignin following the enzymatic/mild acidolysis method (Wu and Argyropoulos 2003; Li et al. 2013) to obtain the enzymatic/mild acidolysis lignin (EMAL).
Procedures for Lignin Hydrothermal Depolymerization
Batch experiments were conducted in a cylindrical autoclave (Yantai Keli chemical equipment Co., Ltd., 300 mL, made of T316 stainless steel) equipped with a pressure gauge, a pressure relief valve, and a stirrer. The autoclave was loaded with 2 g of EMAL and 100 mL of distilled water. The air in the reactor was replaced with nitrogen. The stirring speed was set at 100 rpm. The reactor was heated to between 250 and 350 °C using an external electrical furnace with a constant heating time (25 min), and then it was held at the target temperature for 30 min. The reactor pressure increased and stabilized within 5 min at the target reaction temperatures. The corresponding pressures at 250, 275, 300, 325, and 350 °C were 3.9, 6.0, 8.6, 12.2, and 16.5 MPa, respectively. After the reaction, the electric furnace was removed and the autoclave was rapidly cooled to room temperature using a water bath.
The products were separated and collected, as shown in Fig. 1. The gas products were blown out using high-purity nitrogen (99.99%), which was collected with a gas collecting bag and then analyzed using a gas chromatography with a thermal conductivity detector. The detailed gas analysis method was presented in our previous work. (Lv et al. 2013). The liquid fraction of the products was filtered to separate the water-soluble fraction. Water was added to the water-soluble fraction to reach a final volume of 250 mL. The water-soluble fraction was extracted with ethyl acetate (80 mL 3). The water-insoluble fraction was washed with ethyl acetate until colorless. The resulting insoluble fraction was solid residue (SR). After the SR was dried with anhydrous magnesium sulfate, the ethyl acetate-soluble phase was evaporated in a rotary evaporator to recover the liquid products (LP). The rotary evaporation was set at 40 oC, -0.9 MPa, thereafter, the liquid products and the pre-weighed flat bottom flask was dried in a vacuum drying oven at 40 °C, -0.95 MPa for 24 h to a constant weight. The yields of liquid products were determined by weighing. The content of the EMAL hydrothermolysis product was calculated using Eq. 1,
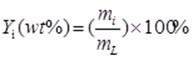
(1)
where mi is the mass of the ith product (g) and mL is the mass of the initial EMAL used for the reaction (g).
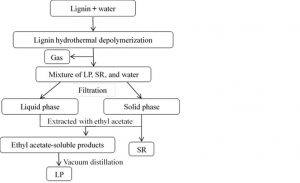
Fig. 1. Schematic for the separation of EMAL products using hydrothermal depolymerization
Analysis Methods
Molecular components of the liquid products were identified using gas chromatography-mass spectrometry (GC-MS; 7890A-5975C, Agilent Technologies, Santa Clara, CA). A HP-INNOWax column (30 m 0.25 mm 0.25 μm, Agilent Technologies, Santa Clara, CA) with He as the carrier gas was used at a constant rate of 1.2 mL·min-1. The sample injection was made in the split ratio of 1:10. The temperature of the column was programmed from 60 to 240 °C and at 10 °C/min-1 after an initial retention time of 2 min and kept at the final temperature for 10 min. The injector and MS detector were maintained at 240 and 230 °C, respectively. The identification of the liquid products was accomplished by comparing the mass spectra obtained with those reported by the National Institute of Standards and Technology Mass Spectral Library. The main monophenols displaying a relatively high content in the liquid products were further quantitated by using external standard method with corresponding the standard substances on a gas chromatograph with a flame ionization detector (GC-FID; 7890A, Agilent Technologies, Santa Clara, CA). The detecting conditions were the same as those used for GC-MS.
The molecular weight distribution of the liquid products was characterized using a liquid chromatograph – mass spectrometer (LC-MS; Bruker maXis impact, Germany) equipped with an ESI source working in the positive mode. Nitrogen was used as the nebulizer gas and was maintained at a flow volume of 4.0 L·min-1 with a nebulizer pressure of 0.3 bar. The charging voltage and capillary voltage were set at 3500 and 2000 V, respectively. The mass range was from m/z 50 to 1500.
The molecular composition of the materials and the solid residues was acquired using a Vario-EL CUBE elemental analyzer (Elementar, Germany).
The chemical structure and functional groups of EMAL and the solid residues were analyzed using spectral analysis with Fourier transform infrared (FTIR) technology (IS 50, Thermo Nicolet, USA). The samples were pressed into a KBr pellet (1:100). The spectra were obtained in the range of 400 to 4000 cm-1 with a resolution of 4 cm-1 and 64 scans for each sample.
Thermogravimetric (TG) analysis of the EMAL and the solid residues was carried out on a thermogravimetric analyzer (TA Q500, TA Instruments, USA). Dynamic TG scans were conducted in the temperature range from 30 to 700 °C at a heating rate of 20 °C/min. The experiments were carried out under a nitrogen atmosphere and at a flow rate of 25 mL·min-1. Approximately 5 to 7 mg of sample was used.
RESULTS AND DISCUSSION
Composition of the EMAL
The raw material, EMAL, contained carbon (59.12%), hydrogen (6.75%), oxygen (33.86%), and nitrogen (0.27%). There was no sulfur detected in the EMAL. The weight-average molecular weight (Mw), the numerical-average molecular weight (Mn), and the polydispersity (Mw/Mn) of the obtained lignin were 20,123 g/mol, 12,372 g/mol, and 1.63, respectively. The composition of EMAL could be illustrated using the empirical formula [C10H13.70O4.30N0.04]n.
Gas, LP, and SR Yields at Different Temperatures
Generally, hydrothermal degradation of the lignin produced three fractions: the liquid product (LP), consisting mostly of phenolic compounds (Zakzeski et al. 2012; Nguyen et al. 2014; Zhu et al. 2015); gaseous products, primarily CO2, CO, H2, CH4, and trace amounts of C2 and C3 hydrocarbons (Yanik et al. 2007, 2008; Sun et al. 2010); and solid residues, including condensed and unconverted lignin.
The gas, LP, and SR yields were obtained at different temperatures, as shown in Fig. 2. There was a modest rise in the gaseous yield when the temperature increased from 250 to 275 °C (Fig. 2). After this, it remained stable between 5% and 10%. The yield of SR declined considerably from 54.18% (at 250 °C) to 30.68% (at 325 °C) and exhibited an upward trend when the temperature ascended further. However, an opposite trend occurred for the LP yield. The LP yield appeared to be maximized (40.38%) at approximately 325 °C. As the reaction temperature was increased to 350 °C, the yield decreased rapidly. During lignin decomposition, the reactions of degradation, dehydration, and condensation occurred simultaneously (Yanik et al. 2007; Sun et al. 2010; Yuan et al. 2010), and there was competition between these reaction processes. The results indicated that the degradation reaction was dominant at a lower temperature (less than 325 °C) because the LP yield increased and SR yield decreased. However, the condensation reaction occurred primarily at higher temperature ranges, where the results showed an increased yield in SR and a decrease in LP. Therefore, the results suggested that lignin depolymerization occurs best at 325 °C

Fig. 2. Yield of the reaction products at various temperatures
Characterization of the Liquid Products from Lignin Hydrothermal Depolymerization
Gas composition
Table 1 illustrates the composition of the gaseous products. The analysis report of the gas composition indicated that the gaseous products were primarily composed of hydrogen (H2), carbon monoxide (CO), carbon dioxide (CO2), and methane (CH4). The H2 yield was minimal, and CH4 was detected when the temperature increased to 300 °C. Other amounts of hydrocarbon increased with increasing temperature. The degradation of carboxyl groups produced primarily CO, CO2, H2, and H2O.
Table 1. Composition of the Gas Products

The main gaseous products from the demethoxylation of guaiacol were CH4 and CO, formed after the homolysis of the -OCH3 bond and hydrogen abstraction from water (Schuchardt et al. 1993). Most of the CO product was expected to come from water-soluble organic compounds, which was consumed through the water-gas shift reaction (CO + H2O = CO2 + H2) and the methanation reaction (CO + 3H2 = CH4 + H2O) (Kruse et al. 2003; Yanik et al. 2007). It should be noted that the methanation reaction is only possible in the presence of a suitable catalyst (Kruse et al. 2003).
Composition of the liquid products
Table 2 shows the results of the qualitative examination of the LP fractions obtained from the EMAL hydrothermal decomposition by GC-MS. The liquid products of hydrothermal depolymerization could be classified as acids, furans, phenols, non-phenolic monocycle arenes, or non-phenolic polycycle arenes. Small quantities of acids and furans resulted from the degradation of carbohydrates, while others were derived from the degradation of C-C and C-O bonds in lignin. Among these products, the phenol content was the greatest, and the main phenolic products were phenol,2-methoxy- ; phenol,2-methoxy-4-methyl-; phenol,2-methoxy-4-ethyl-; vanillin; ethanone,1-(4-hydroxy-3-methoxyphenyl)-; and 2-propanone,1-(4-hydroxy-3-methoxy phenyl)-. The products were quantified using GC-FID, and the results are illustrated in Fig. 3. An increase in the reaction temperature resulted in higher contents of phenol,2-methoxy-; phenol,2-methoxy-4-methyl-; ethanone,1-(4-hydroxy-3-methoxyphenyl)-; and 2-propanone,1-(4-hydroxy-3-methoxyphenyl)-; and a maximum in the product yields was reached at 325, 325, 325, and 300 °C, respectively. When the temperature increased to 350 °C, the yields decreased because of secondary decomposition. The yield of phenol, 2-methoxy-4-methyl- increased notably from 0.33 to 5.38 mg·g-1. Conversely, the formation of vanillin was reduced considerably from 7.92 to 1.23 mg·g-1 when the temperature rose above 325 °C. In the lignin hydrothermal degradation process, the cleavage of β-O-4, α-O-4, and 4-O-5 bonds, the main bonds connecting structure units, can produce phenol, 2-methoxy-. This is a possible explanation for why the yield of phenol, 2-methoxy- was relatively high in the LP medium. Perhaps phenolic products containing unsaturated functional group (e.g., C=C and C=O) were products of initial lignin depolymerization. When the reaction temperature increased, this unsaturated functional group may have collapsed, leading to an increase in the number of saturated hydrocarbons, such as phenol, 2-methoxy-4-methyl-.

Fig. 3. The effect of hydrothermal temperature on the yield of selected components in the LP
Table 2. Identification of Compounds in the LP by GC-MS Analysis at Various Temperatures

a Value in parentheses show the number of the detected compounds in a specific category
The appearance of catechol, phenol, and phenol substituted by alkyl, and the reduction of guaiacol indicated that demethylation and demethoxylation occurred as secondary reactions at 350 °C (Ren et al. 2012).
Molecular weight distribution of the liquid products
The GC-MS and GC-FID analyses were used only to detect the volatile fractions at certain experimental conditions in the liquid products. There were still many substances not checked. Therefore, the molecular weight distribution of the liquid products collected at different temperatures was further investigated using LC/ESI-MS, and the results are presented in Fig. 4. For Cunninghamia lanceolata, the main prototype lignin subunit was phenylpropane guaiacol (Li and Wu 2014), with a molecular weight of 166 g·mol−1. It was observed in Fig. 4 that the molecular weight distribution pattern was very different at varying temperatures, indicating that the temperature was one of the main factors involved in the hydrothermal depolymerization of lignin. Most of the liquid products obtained at 250 °C were dimers and trimers; however, at a higher temperature, the content of the liquid products with a larger molecular weight was enhanced. This phenomenon could have possibly resulted from the condensation of the lignin degradation products as a secondary reaction at a relatively high liquid-phase concentration and temperature.

Fig. 4. LC/ESI-MS analysis of the LP at different temperatures
Properties of the Solid Residues from Lignin Hydrothermal Depolymerization
Elementary analysis of solid residues
The contents of the organic elements (C, H, N, and S) in the EMAL and their solid residues are presented in Table 3. The content of oxygen was calculated by difference. It was observed that the C, H, and N contents of the solid residues obtained at different temperatures differed considerably from the raw material, which suggested changes in their structures. The carbon content was notably enhanced after lignin hydrothermal depolymerization, and peaked at 275 °C. Conversely, the oxygen content of the solid residues was lower than the EMAL and reached a maximum at 350 °C. The high heating values (HHV) of the residues are listed in Table 3. These were calculated using the Dulong formula (Yuan et al. 2009). The HHV initially increased with increasing temperature and peaked (27.63 MJ/kg) at 275 °C; however, the HHV decreased subsequently when the temperature increased from 275 to 350 °C.
Table 3. Elemental Analysis of the Solid Residues

FTIR spectral analysis of the residues
To reveal the functional groups and changes in chemical structure from the lignin hydrothermal decomposition process, FTIR analyses of the material and the SR were carried out. Their FTIR spectra are shown in Fig. 5. The differences in the FTIR spectra were pronounced. The wavenumbers 3437, 1267, and 1032 cm-1 corresponded to -OH stretching, =C-O-R stretching at the guaiacyl units, and C-O bending from the aromatic ring, respectively. The signals at these bands weakened with rising temperatures, which suggested that the lignin structure was gradually damaged, and the condensation reaction of the lignin occurred. The band typical for -CH3,-CH2,-CH bending was diminished at 2935 and 2839 cm-1. The demethylation and dealkylation reactions occurred during the hydrothermal depolymerization of lignin. The absorption band centered at 1699 cm-1 originated from conjugated carbonyl. Its absence at 325 °C indicated that decarbonylation occurred and that CO2 was present in the gases produced. The appearance of the absorption band could be ascribed to the carbonylation of EMAL at 250 oC under hydrothermal condition. The peaks at 1085 and 1032 cm-1 gradually disappeared, suggesting the loss of the ether band in the residues. The FTIR spectrums of the solid residues and EMAL were similar within the reaction temperature range of 250 to 300 °C, which indicated that their functional types and chemical structures were similar. However, when the reaction temperature increased to 325 °C, there was a relatively large change in the absorption spectrum. This suggested that the solid residues were mostly composed of phenolic biochar.

Fig. 5. FTIR spectra of EMAL and solid residues


Fig. 6. TGA and DTG distributions of the solid residues
To study the changes in thermal properties of the solid residues, TGA and first derivation thermogravimetric (DTG) analyses were performed; the results are shown in Fig. 6. In the TG curve, the solid residue yield after 700 °C increased as the hydrothermal reaction temperature increased. As shown by the DTG curve, the initial mass loss at approximately 90 °C was attributed to the moisture content of the samples. At 250 °C, there were two main weight loss peaks: at 375 and 413 °C. By contrast, the other four had only one peak and it was shifted to higher temperatures with the increasing hydrothermal reaction temperature. These results illustrated that the solid residues of lignin after the hydrothermal reaction exhibited a higher thermal stability than the raw material and that the thermal stability increased as the hydrothermal reaction temperature increased.
CONCLUSIONS
- The hydrothermal depolymerization of Cunninghamia lanceolata enzymatic/mild acidolysis lignin (EMAL) was performed in subcritical water. The yield of the liquid products peaked at a preparation temperature of 325 °C.
- The liquid products were primarily phenol and phenol, 2-methoxy-. The highest monomer content peaked at 325 °C.
- Lignin could be broken down by the simultaneous processes of hydrothermal depolymerization and condensation at 250 °C.
- When the reaction temperature was increased to 325 °C, the solid residues mostly consisted of phenolic biochar.
ACKNOWLEDGMENTS
The authors gratefully acknowledge the financial support of the National Basic Research Program of China (2013CB228101, 973 program), the State Natural Sciences Foundation (31270635 & 21176095), and the Fundamental Research Funds for the Central Universities (2014ZP14).
REFERENCES CITED
Fang, Z., Sato, T., Smith, R. L., Inomata, H., Arai, K., and Kozinski, J. A. (2008). “Reaction chemistry and phase behavior of lignin in high-temperature and supercritical water,” Bioresour. Technol. 99(9), 3424-3430. DOI: 10.1016/j.biortech.2007.08.008
García, A., Toledano, A., Serrano, L., Egües, I., Gonzáles, M., Marin, F., and Labidi, J. (2009). “Characterization of lignins obtained by selective precipitation,” Sep. Purif. Technol. 68(2), 193-198. DOI: 10.1016/j.seppur.2009.05.001
Guerra, A., Filpponen, I., Lucia, L. A., and Argyropoulos, D. S. (2006). “Comparative evaluation of three lignin isolation protocols for various wood species,” J. Agric. Food Chem. 54(26), 9696-9705. DOI: 10.1021/jf062433c
Ikeda, T., Holtman, K., Kadla, J. F., Chang, H.-M., and Jameel, H. (2002). “Studies on the effect of ball milling on lignin structure using a modified DFRC method,” J. Agric. Food Chem. 50(1), 129-135. DOI: 10.1021/jf010870f
Kanetake, T., Sasaki, M., and Goto, M. (2007). “Decomposition of a lignin model compound under hydrothermal conditions,” Chem. Eng. Technol. 30(8), 1113-1122. DOI: 10.1002/ceat.200700066
Kruse, A., Henningsen, T., Sinag, A., and Pfeiffer, J. (2003). “Biomass gasification in supercritical water: Influence of the dry matter content and the formation of phenols,” Ind. Eng. Chem. Res. 42(16), 3711-3717. DOI: 10.1021/ie0209430
Li, J. B., Wu, S. B., and Li, X. H. (2013). “Chemical structure and thermochemical properties of enzymatically acidolyzed lignins from soft and hard wood,” BioResources 8(4), 5120-5132. DOI: 10.15376/biores.8.4.5120-5132
Li, X. H., and Wu, S. (2014). “Chemical structure and pyrolysis characteristics of the soda-alkali lignin fractions,” BioResources 9(4), 6277-6289. DOI: 10.15376/biores.9.4.6277-6289
Lv, G., Wu, S., Yang, G., Chen, J., Liu, Y., and Kong, F. (2013). “Comparative study of pyrolysis behaviors of corn stalk and its three components,” Journal of Analytical and Applied Pyrolysis 104(11), 185-193. DOI:10.1016/j.jaap.2013.08.005
Nguyen, T. D., Maschietti, M., Belkheiri, T., Amand, L. E., Theliander, H., Vamling, L., Olausson, L., and Andersson, S. I. (2014). “Catalytic depolymerisation and conversion of kraft lignin into liquid products using near-critical water,” J. Supercrit. Fluid. 86, 67-75. DOI: 10.1016/j.supflu.2013.11.022
Okuda, K., Ohara, S., Umetsu, M., Takami, S., and Adschiri, T. (2008). “Disassembly of lignin and chemical recovery in supercritical water and p-cresol mixture: Studies on lignin model compounds,” Bioresour. Technol. 99(6), 1846-1852. DOI: 10.1016/j.biortech.2007.03.062
Ren, M. M., Li, H. S., Zhang, M. H., Wang, G. Q., and Sun, Y. P. (2012). “Research progress on the application of lignin,” Polym. Bullet. 8, 44-49. DOI: 10.14028/j.cnki.1003-3726.2012.08.020
Sasaki, M., and Goto, M. (2008). “Recovery of phenolic compounds through the decomposition of lignin in near and supercritical water,” Chem. Eng. Process. 47(9-10), 1609-1619. DOI: 10.1016/j.cep.2007.09.001
Sasaki, M., and Goto, M. (2009). “Conversion of biomass model compound under hydrothermal conditions using batch reactor,” Fuel 88(9), 1656-1664. DOI: 10.1016/j.fuel.2009.02.028
Sasaki, M., and Goto, M. (2011). “Thermal decomposition of guaiacol in sub-and supercritical water and its kinetic analysis,” J. Mater. Cycles Waste. Manag. 13(1), 68-79. DOI: 10.1007/s10163-010-0309-6
Schuchardt, U., Rodrigues, J. A., Cotrim, A. R., and Costa, J. M. (1993). “Liquefaction of hydrolytic eucalyptus lignin with formate in water, using batch and continuous-flow reactors,” Bioresour. Technol. 44(2), 123-129. DOI: 10.1016/0960-8524(93)90185-E
Sun, P., Heng, M., Sun, S., and Chen, J. (2010). “Direct liquefaction of paulownia in hot compressed water: Influence of catalysts,” Energy 35(12), 5421-5429. DOI: 10.1016/j.energy.2010.07.005
Toledano, A., García, A., Mondragon, I., and Labidi, J. (2010). “Lignin separation and fractionation by ultrafiltration,” Sep. Purif. Technol. 71(1), 38-43. DOI: 10.1016/j.seppur.2009.10.024
Toledano, A., Serrano, L., and Labidi, J. (2012). “Organosolv lignin depolymerization with different base catalysts,” J. Chem. Technol. Biotech. 87(11), 1593-1599. DOI: 10.1002/jctb.3799
Wu, S., and Argyropoulos, D. S. (2003). “An improved method for isolating lignin in high yield and purity,” J. Pulp Pap. Sci. 29(7), 235-240.
Yanik, J., Ebale, S., Kruse, A., Saglam, M., and Yüksel, M. (2007). “Biomass gasification in supercritical water: Part 1. Effect of the nature of biomass,” Fuel 86(15), 2410-2415. DOI: 10.1016/j.fuel.2007.01.025
Yanik, J., Ebale, S., Kruse, A., Saglam, M., and Yüksel, M. (2008). “Biomass gasification in supercritical water: II. Effect of catalyst,” Int. J. Hydrogen Energ. 33(17), 4520-4526. DOI: 10.1016/j.ijhydene.2008.06.024
Yuan, X. Z., Tong, J. Y., Zeng, G. M., Li, H., and Xie, W. (2009). “Comparative studies of products obtained at different temperatures during straw liquefaction by hot compressed water,” Energy Fuels 23(6), 3262-3267. DOI: 10.1021/ef900027d
Yuan, Z., Cheng, S., Leitch, M., and Xu, C. (2010). “Hydrolytic degradation of alkaline lignin in hot-compressed water and ethanol,” Bioresour. Technol. 101(23), 9308-9313. DOI: 10.1016/j.biortech.2010.06.140
Zakzeski, J., Jongerius, A. L., Bruijnincx, P. C. A., and Weckhuysen, B. M. (2012). “Catalytic lignin valorization process for the production of aromatic chemicals and hydrogen,” ChemSusChem 5(8), 1602-1609. DOI: 10.1002/cssc.201100699
Zhu, Z., Toor, S. S., Rosendahl, L., Yu, D., and Chen, G. (2015). “Influence of alkali catalyst on product yield and properties via hydrothermal liquefaction of barley straw,” Energy 80(1), 284-292. DOI: 10.1016/j.energy.2014.11.071
Article submitted: July 7, 2015; Peer review completed: September 26, 2015; Revised version received: October 11, 2015; Accepted: October 14, 2015; Published: November 5, 2015.
DOI: 10.15376/biores.11.1.21-32
