Abstract
Spent mushroom substrate (SMS) is a noxious byproduct of the mushroom industry. The aim of this work was to convert SMS into organic-mineral micronutrient (Zn(II), Mn(II), and Cu(II)) fertilizer via biosorption and examine the effect of its application in field tests on maize compared to commercial reference micronutrient fertilizer. Crop yield and crop quality were assessed, and multielemental analysis of grains was conducted for the evaluation of the fertilization effect on maize grains and to assess bioavailability of nutrients from fertilizers. Grain yield for maize treated with micronutrients delivered with SMS was noticeably higher (11.5%) than the untreated group and the NPK (nitrogen, phosphorus, potassium) fertilizer treated only group (2.8%). Bioavailability (TF) of micronutrients from SMS were comparable with reference micronutrient fertilizer (7% Zn, 4% Mn, and 2.3% Cu). The new product has the potential to be used as a micronutrient fertilizer. Satisfactory results of grain yield (6.4 Mg ha-1), high content of micronutrients (Zn 1.6%, Mn 1.2%, and Cu 1.8%), and macronutrients (P 1.0%, S 3.1%, Ca 8.2%, and K 0.2%) were observed. The bioavailability suggests that enriched SMS could be a good alternative to fertilizers in the present market.
Download PDF
Full Article
Using Spent Mushroom Substrate as the Base for Organic-Mineral Micronutrient Fertilizer – Tests on Maize
Łukasz Tuhy,* Mateusz Samoraj, Zuzanna Witkowska, Radosław Wilk, and Chojnacka
Spent mushroom substrate (SMS) is a noxious byproduct of the mushroom industry. The aim of this work was to convert SMS into organic-mineral micronutrient (Zn(II), Mn(II), and Cu(II)) fertilizer via biosorption and examine the effect of its application in field tests on maize compared to commercial reference micronutrient fertilizer. Crop yield and crop quality were assessed, and multielemental analysis of grains was conducted for the evaluation of the fertilization effect on maize grains and to assess bioavailability of nutrients from fertilizers. Grain yield for maize treated with micronutrients delivered with SMS was noticeably higher (11.5%) than the untreated group and the NPK (nitrogen, phosphorus, potassium) fertilizer treated only group (2.8%). Bioavailability (TF) of micronutrients from SMS were comparable with reference micronutrient fertilizer (7% Zn, 4% Mn, and 2.3% Cu). The new product has the potential to be used as a micronutrient fertilizer. Satisfactory results of grain yield (6.4 Mg ha-1), high content of micronutrients (Zn 1.6%, Mn 1.2%, and Cu 1.8%), and macronutrients (P 1.0%, S 3.1%, Ca 8.2%, and K 0.2%) were observed. The bioavailability suggests that enriched SMS could be a good alternative to fertilizers in the present market.
Keywords: Spent mushroom substrate (SMS); Biomass utilization; Field tests; Micronutrient fertilizer; Micronutrients
Contact information: Department of Advanced Material Technologies, Faculty of Chemistry, Wrocław University of Technology, Smoluchowskiego 25, 50-372 Wrocław, Poland;
* Corresponding author: lukasz.tuhy@pwr.edu.pl
INTRODUCTION
Spent mushroom substrate (SMS) is a byproduct of the mushroom industry and is often treated as waste. It was calculated that the production of one kilogram of mushrooms generates about five kilograms of SMS (Kapu et al. 2007; Finney et al. 2012). On many farms, SMS is disposed of as noxious waste material (Shi et al.2014). SMS is regarded as biomass because of its high organic matter content, especially lignocelluloses (Garrido et al. 2005).
With the development of the mushroom industry, the amount of SMS has increased, and more attention has been paid to the utilization of this waste in such a way that it becomes a valuable product (Phan and Sabaratnam 2012). The problem with the storing, disposing, and/or utilization of SMS has been reported in Poland, which is the third largest producer of mushrooms in the world (250 to 300 thousand tons per year) (Kalembasa and Becher 2012).
Among many different characteristics and possible applications, SMS was also shown to be a source of enzymes, enabling its use in animal nutrition, energy production, or even bioremediation (Phan and Sabaratnam 2012). There are also some reports describing the use of SMS in agriculture to increase the organic content of soil and to improve its structure. The positive effect of the application of SMS as an organic fertilizer instead of manure was demonstrated in tests carried out on pineapple (Orluchukwu and Adedokun 2014). Eudoxie and Alexander (2011) showed that SMS can be used as a media replacement for commercial peat. The effect of the fertilization with SMS on seedlings of tomato, cucumber, and watermelon have been described by Zhan and Duan (2012). The application of SMS in agriculture can be preceded by different transformations improving its fertilizing properties. Zhu and co-workers (2012) examined the use of SMS in the production of biofertilizer with the use of phosphate solubilizing microorganisms. SMS was also shown to be characterized by good sorption properties (Chen et al. 2005). The many organic compounds and functional groups found on the surface of agricultural wastes such as SMS make waste biomass a good biosorbent (Dhankhar and Hooda 2011; Putra et al. 2014; Toptas et al. 2014).
Biosorption is the ability of biomaterials to bind molecules or ions in water solutions (Qi and Aldrich 2008; Fomina and Gadd 2014). It is a metabolically-independent process, which is based on a variety of mechanisms, such as ion exchange, covalent binding, adsorption, microprecipitation, and Van der Waals attraction (Kratochvil and Volesky 1998; Witek-Krowiak 2012). Furthermore, biosorption is a cost-effective process ready to be used in industry (Abdolali et al.2014). The number of applications in which biosorption could be used is growing. Biosorption is widely described as a removal tool for heavy metals and dyes from wastewater (Sulaymon et al. 2013; Hadavifar et al. 2014; Jain et al. 2014). There are also some publications describing the use of biosorption in the production of micronutrient feed additives and its efficient application in animal nutrition (Michalak et al. 2011; Saeid et al. 2013; Witkowska et al.2014). Micronutrients are engaged in many biochemical pathways in living organisms, and deficiency of these metals can affect proper cell functionality (Hsieh et al. 2013).
Micronutrient malnutrition can cause serious diseases, not only in plants and animals, but also in humans. Micronutrient deficiency is the main reason of so-called hidden hunger, characterized by the lack of the appropriate balance of nutrients (Murgia et al. 2012). One of the methods for combating micronutrient deficiency is micronutrient fertilization (Muller et al. 2014). Popular micronutrient fertilizers are mainly cheap inorganic salts characterized by high leachability from soil to groundwater and expensive and non-biodegradable organic chelates (Murakami et al. 2003; Jie et al. 2008). According to this, there is a need for new fertilizer materials that are cheap, biodegradable, and have high bioavailability of nutrients (Michalak et al. 2013). The compilation of biosorption properties of SMS with the potential of its use in agriculture make it a promising micronutrient fertilizer or fertilizer component constituting a tool to overcome micronutrient deficiency in soils and plants.
The aim of the present work was to utilize lignocellulosic biomass of SMS in a converted form as an organic-mineral micronutrient fertilizer via biosorption and examine the effect of its application in field tests on maize in comparison with commercial micronutrient fertilizer.
EXPERIMENTAL
Micronutrient Bio-components Production
Micronutrient fertilizer bio-components were produced in a biosorption process. For the biosorption experiments, SMS delivered by the Hajduk mushroom farm (Poland) was used. SMS used in experiments was the spent growth medium after the cultivation of Agaricus bisporus. The biosorption of zinc(II), copper(II), and manganese(II) ions by biological material was conducted in a fixed bed column reactor (200 L) separately for each micronutrient for 6 h. The concentration of Zn(II) (ZnSO4∙7H2O; POCH, Poland), Cu(II) (CuSO4∙5H2O; POCH, Poland), and Mn(II) (MnSO4∙1H2O; POCH, Poland) ions in the solutions was 500 mg/L for each process. The pH was held at 5 throughout the biosorption process. The biosorption process was conducted at 25 °C. In each process 20 kg of biosorbent was used. After each batch, the suspension was transferred to a filter press (MONTECH PKF 500, Poland) for biomass recovery. The final product was dried in an industrial dryer (Hajnowka, Poland) at 50 °C for 24 h. The content of elements in the enriched biomass was examined by ICP–OES after mineralization.
Field Trials
Field trials were conducted on maize (KOSMO 230) at the Plant Breeding and Acclimatization Experimental Station in Olesnica Mala (Lower-Silesia, southwestern Poland). The characteristics of the soil were as follows: sandy loam, IIIb quality class, 2.2% organic matter, and pH 7.2. The experimental area was divided into 21 m2 plots. The randomization of blocks is presented in Fig. 1.
Fig. 1. Experimental groups arrangement on the field ( “-“ = empty plot, “1” = SMS+ Zn, Mn, Cu; “2” = untreated plot; “3” = NPK; “4” = NPK + Zn, Mn, Cu)
The interval between rows of plants was 75 cm, and 16 cm between individual plants. Planting density was 85000 pcs of corn seeds/ha. Each combination was carried in 4 replications. The experiment was conducted during 6 months (from May to October). The average temperature was 15.2 ±4.3 °C and the average month total rainfall was 68.7 mm.
Fertilization
For the experiment, bio-components with zinc, manganese, and copper, based on the biomass of SMS, were produced. For each prepared combination of micronutrient fertilizer bio-component, an additional NPK (nitrogen, phosphorus, potassium) fertilizer, Polifoska 4 NPK (MgS) (4% of N, 12% P, 32% K, 2% Mg, and 9% S), was applied (SMS + Zn, Mn, Cu). The fertilizer was delivered by Grupa Azoty Z. Ch. “Police” S.A., Poland. To compare fertility results, 3 control combinations were used; untreated (UNTREATED), NPK(MgS) (NPK), and NPK(MgS) with micronutrients (technical salts of Zn, Cu, Mn; POCH, Poland) (NPK + Zn, Mn, Cu). The micronutrients ratio in applied micronutrient bio-components and NPK(MgS) fertilizer with micronutrients was 1:0.4:0.2 for Zn, Mn, and Cu, respectively. The quantitative description of fertilization within tested combinations on the field is listed in Table 1. The dosages of the commercial products were in accordance with the manufacturer’s recommendation for maize fertilization. Doses of bio-components are equivalent to commercial products. During the experiment, all plant parameters were examined according to the guidelines EPPO PP 1/144 (2) (2012), EPPO PP 1/135 (4) (2014), EPPO PP 1/152 (4) (2012), and EPPO PP 1/181 (4) (2012) documents approved by European and Mediterranean Plant Protection Organization (EPPO).
Table 1. Fertilizer Dosages
Mineralization
Each material (1 g) was digested with nitric acid, 69% m/m (5 mL) spectrally pure, (Suprapur, Merck, USA) in teflon bombs in a Milestone Start D (USA) microwave system. Parameters of the mineralization process were matched to assure complete digestion of samples. Samples were diluted 10 times with ultrapure water (Millipore Simplicity, Germany) to perform multielemental ICP-OES analysis.
Multielemental ICP-OES Analysis
The concentration of elements in digested biomass was determined by ICP–OES (inductively coupled plasma optical emission spectrometry) Varian-Vista MPX (Australia). Samples were served with ultrasonic nebulizer CETAC U5000AT+. The analyses were carried out in a laboratory accredited by the Polish Centre of Accreditation (PCA) according to PN-EN ISO/IEC 17025:2005. Quality assurance of the test results was achieved by using Combined Quality Control Standard from ULTRA SCIENTIFIC (USA). All samples were analyzed in three repeats (results of analyses were arithmetic mean, and the relative standard deviation was <5%).
Statistical Analysis
The results were elaborated statistically by Statistica (version 10, Poland), and descriptive statistics (means, standard deviations) were reported. Normality of distribution of experimental results was assessed by the Shapiro–Wilk test. On this basis, the (RIR) Tukey statistical test for the investigation of the significance of differences between the groups was selected, which compares all pairs of means following one-way ANOVA. Results were considered significantly different when p<0.05
RESULTS AND DISCUSSION
Multi-elemental Analysis of Enriched Biomass
Table 2 shows multi-elemental contents of new bio-components, from ICP–OES.
Table 2. Multi-element Content of New Bio-components
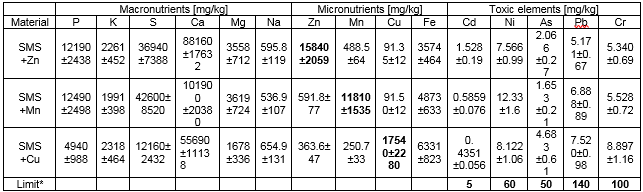
* Act of Fertilizer and Fertilization, 18 June 2008, approved by Polish Ministry of Agriculture and Rural Development.
The contents of micronutrients, after the biosorption process were 15840 mg/kg, 11180 mg/kg, and 17540 mg/kg for zinc, manganese, and copper, respectively. SMS was also shown to be a rich source of Fe (0.5%). In addition to micro-elements, biomass enriched viabiosorption contained macronutrients in high concentrations, especially P (1.0%), S (3.1 %), Ca (8.2%), and K (0.2%). Because of the organic character of the biomass micro- and macronutrients content, SMS can be classified as an organic-mineral fertilizer. Toxic elements (Cd, Ni, As, Pb, and Cr) were far below than the admissible limit, defined in the Act of Fertilizer and Fertilization (2008), approved by Polish Ministry of Agriculture and Rural Development.
Plant Parameters
Among parameters describing the quality of plant yield, plant vigour, and plant height, the number of plants and cob number were examined and presented in Table 3.
Table 3. Qualitative Description of Maize
*vigour on a 0 to 10 scale: 0 = plant death, 5 = optimum vigour (Untreated), 10 = most vigorous plants;
Tukey test: statistically significant differences for a given element between materials, a,b…(p<0.05)
No phytotoxicity effect was observed. Neither was it observed for the application of SMS with micronutrients, nor for mineral fertilizer with micronutrients.
The quantitative comparison of crop yield between groups is presented in Table 4. Obtained grain yield was comparable for plants fertilized with commercial micronutrient fertilizer (NPK + Zn, Mn, Cu) and SMS (SMS+ Zn, Mn, Cu). The highest grain yield was obtained for plants fertilized with micronutrient fertilized groups (SMS+ Zn, Mn, Cu and NPK + Zn, Mn, Cu yielded 6.4 Mg/ha and 6.6 Mg/ha, respectively).
Table 4. Yield Properties
*at 15% moisture
Micronutrient Content
The micronutrient content in maize grain after field trials was determined by ICP-OES, and the results are presented in Table 5. The values of transfer factor (TF) constituting a nutrient mass in grain to mass of delivered nutrient ratio (Tuhy et al. 2013) was calculated with the following Eq. 1:
(1)
The results characterizing the availability of nutrients to plants were compared and are presented in Table 5.
Table 5. Multi-element Content of Maize Grain
Tukey test: statistically significant differences for a given element between materials, A,B…(p<0.1)
The highest mass of zinc, manganese, and copper in the grain was observed for SMS+ Zn, Mn, Cu (386 mg Zn, 92.4 mg Mn, 25.6 mg Cu) and for the reference commercial micronutrient fertilizer – NPK + Zn, Mn, Cu (392 mg Zn, 95.2 mg Mn, 25.6 mg Cu). The highest content of zinc and copper in grains was observed for SMS+ Zn, Mn, Cu, and it was 10.5% and 14.0% higher than groups treated with only NPK fertilizer. The highest transfer factor of Zn(II) and Cu(II) was obtained for SMS enriched with micronutrients.
SMS can be a promising new fertilizer material with advantageous properties, such as the preferred C:N ratio, assimilable forms of nutrients, and high content of organic matter (Garrido 2012). SMS naturally contains macronutrients in high concentrations, especially P (1.0%), S (3.1 %), Ca (8.2%), and K (0.2%) (Table 2). The optimal range for organic composts varies from 0.6% to 0.9% for P, and from 0.2% to 0.5% for K (Ribas et al. 2009). A high concentration of Ca(II) ions was also observed by other researchers (Wiśniewska-Kadżajan and Jankowski 2013). Application of SMS improves soil fertility (Medina et al. 2012). Toxic elements (Cd, Ni, As, Pb, and Cr) were far lower than the admissible limit (Table 2). Wiśniewska-Kadżajan and Jankowski (2013) also reported low heavy metal content in SMS+ Zn, Mn, Cu.
Obtained grain yield with SMS+ Zn, Mn, Cu treated plots was noticeably higher (11.5%) than the untreated group and the treated NPK only group (2.8%). Comparable results were obtained for SMS+ Zn, Mn, Cu and reference micronutrient fertilizer (NPK + Zn, Mn, Cu), 6.4 and 6.6 Mg/ha, respectively (Table 4). These findings indicated that SMS has the potential to be used as a micronutrient fertilizer for slow release and also as a soil-conditioning and bioremediation agent, confirming the results obtained by Ribas et al.(2009). Zinc content in grains, for plants treated with enriched SMS, was higher by about 10.5% than for plants fertilized only by NPK fertilizer. Lungu and coworkers (2011), in similar tests on maize, obtained comparable results (6% to 15%) with soil application of 50 kg/ha of ZnSO4∙7H2O (11.4 kg/ha of pure Zn). In other work, Menzeke and coworkers (2014) obtained 7.2% to 18% increase of Zn in grain of maize with cattle manure application with Zn.
Bioavailability (TF) of micronutrients from SMS were comparable with the reference micronutrient fertilizer (NPK + Zn, Mn, Cu) (for Zn and Cu, bioavailability was slightly higher) (Table 5). No statistically significant differences in micronutrient content of grain were found between the SMS + Zn, Mn, Cu and commercial micronutrient fertilizer (Table 5). No statistically significant differences were found in the bioavailability (TF) of nutrients between commercial micronutrient fertilizer and bio-preparations.
Similar field trials on maize were conducted with natural SMS, and plant yield was significantly higher in SMS amended plots. Also, a good quality of plant yield was observed in the experiment (Wuest et al. 1995). Other researchers used SMS for the fertilization of tomato, courgette, pepper (Medina et al. 2009), and spinach (Ahlawat and Sagar 2007). Observed effects were comparable to traditional fertilization.
Soil application is a promising strategy for the recycling of SMS. In comparison with other organic fertilizers (urban wastes and animal manure), SMS has similar organic matter and higher macronutrient concentrations. The effectiveness of SMS as an organic fertilizer has been positively evaluated (Medina et al. 2012). Satisfactory results of grain yield, content of micro- and macronutrients, and their high bioavailability suggests that SMS enriched with micronutrients could be a good alternative for fertilizers present on the market. Utilization of SMS into an organic-mineral micronutrient fertilizer is an alternative way of using spent mushroom compost for micro- and macronutrient fertilization. Nutrients are present in sufficient quantity and in the available form (Ahlawat and Sagar 2007). This shows that the use of SMS with micronutrients via biosorption contributes to disposal in an environmentally friendly way and has a positive effect on plant fertilization.
CONCLUSIONS
- Spent mushroom substrate (SMS), a waste raw material, can be converted into micronutrient fertilizer via a biosorption process.
- The fertilization efficiency of the new preparation was comparable to commercial reference micronutrient fertilizer.
- SMS improved soil structure, quality, and its sorption capacity. It is also characterized by preferred ratios of macronutrients that are essential to plants in cultivation.
- Micronutrients delivered with new bio-preparations are characterized by high bioavailability (high values of TF), comparable to commercial products.
- SMS enriched with micronutrients in the biosorption process can be treated as an alternative to traditional micronutrient fertilization with the direct use of inorganic salts.
ACKNOWLEDGMENTS
This work was financed by Polish National Science Center, project No UMO-2012/05/E/ST8/03055. Publication was supported by the Wroclaw Centre of Biotechnology under The Leading National Research Centre (KNOW) programme for the years 2014 to 2018.
REFERENCES CITED
Abdolali, A., Guo, W., Ngo, H., Chen, S., Nguyen, N., and Tung, K. (2014). “Typical lignocellulosic wastes and by-products for biosorption process in water and wastewater treatment: A critical review,” Biores. Technol. 160, 57-66. DOI:10.1016/j.biortech.2013.12.037
Ahlawat, O., and Sagar, M. (2007). Management of Spent Mushroom Substrate, National Research Centre for Mushrooms, Solan (ed.), Indian Council of Agricultural Research.
Chen, G., Zeng, G., Tu, X., Huang, G., and Chen, Y. (2005). “A novel biosorbent: Characterization of the spent mushroom compost and its application for removal of heavy metals,” J. Environ. Sci. 17(5), 756-760.
Dhankhar, R., and Hooda, A. (2011). “Fungal biosorption: An alternative to meet the challenges of heavy metal pollution in aqueous solutions,” Environ. Technol. 32(5), 467-491. DOI:10.1080/09593330.2011.572922
EPPO PP 1/135 (4) (2014). “Phytotoxicity assessment,” Efficacy evaluation of plant protection products, Paris, France
EPPO PP 1/144 (2) (2012). “Reduction of lodgings in cereals and maize,” Efficacy evaluation of of plant growth regulators, Paris, France
EPPO PP 1/152 (4) (2012). “Design and analysis of efficacy evaluation trials,” Efficacy evaluation of plant protection products, Paris, France
EPPO PP 1/181 (4) (2012). “Conduct and reporting of efficacy evaluation trials, including good experimental practice,” Efficacy evaluation of plant protection products, Paris, France
Eudoxie, G., and Alexander, I. (2011). “Spent mushroom substrate as a transplant media replacement for commercial peat in tomato seedling production,” J. Agric. Sci. 3(4), 41-49. DOI:10.5539/jas.v3n4p41
Finney, K., Ryu, C., Sharifi, V., and Swithenbank, J. (2009). “The reuse of spent mushroom compost and coal tailings for energy recovery: comparison of thermal treatment technologies,” Biores. Technol. 100(1), 310-315. DOI:10.1016/j.biortech.2008.05.054
Fomina, M., and Gadd, G. (2014). “Biosorption: current perspectives on concept, definition and application,” Biores. Technol. 160, 3-14. DOI:10.1016/j.biortech.2013.12.102
Garrido, R., Ruiz-Felix, M., and Satrio, J. (2012). “Effects of hydrolysis and torrefaction on pyrolysis product distribution of spent mushroom compost (SMC),” Int. J. Environ. Pollut. Rem. 1(1), 98-103. DOI:10.11159/ijepr.2012.014
Hadavifar, M., Bahramifar, N., Younesi, H., and Li, Q. (2014). “Adsorption of mercury ions from synthetic and real wastewater aqueous solution by functionalized multi-walled carbon nanotube with both amino and thiolated groups,” Chem. Eng. J. 237, 217-228. DOI:10.1016/j.cej.2013.10.014
Hsieh, S., Castruita, M., Malasarn, D., Urzica, E., Erde, J., Page, M., Yamasaki, H., Casero, D., Pellegrini, M., Merchant, S.S., and Loo, J.A. (2013). “The proteome of copper, iron, zinc, and manganese micronutrient deficiency in Chlamydomonas reinhardtii,” Mol. Cell. Proteom. 12(1), 65-86. DOI:10.1074/mcp.M112.021840
Jain, R., Mody, K., Keshri, J., and Jha, B. (2014). “Biological neutralization and biosorption of dyes of alkaline textile industry wastewater,” Mar. Pollut. Bull. 84(1), 83-89. DOI:10.1016/j.marpolbul.2014.05.033
Jie, M., Raza, W., Xu, Y., and Shen, Q. (2008). “Preparation and optimization of amino acid chelated micronutrient fertilizer by hydrolyzation of chicken waste feathers and the effects on growth of rice,” J. Plant. Nutt. 31(3), 571-582. DOI:10.1080/01904160801895092
Kalembasa, D., and Becher, M. (2012). “Speciation of carbon and selected metals in spent mushroom substrates,” J. Elem. 17(3), 409-419. DOI:10.5601/jelem.2012.17.3.04
Kapu, N., Manning, M., Hurley, T., Voigt, J., Cosgrove, D., and Romaine C. (2012). “Surfactant-assisted pretreatment and enzymatic hydrolysis of spent mushroom compost for the production of sugars,” Biores. Technol. 114, 399-405. DOI:10.1016/j.biortech.2012.02.139
Kratochvil, D., and Volesky, B. (1998). “Advances in the biosorption of heavy metals,” Trends. Biotechnol. 16(7), 291-300. DOI:10.1016/S0167-7799(98)01218-9
Lungu, O., Simunji, S., and Cakmak, I. (2011). “Effects of soil and foliar applications of zinc on grain zinc concentrations of maize, sorghum and wheat in Zambia,” INTSORMIL 1, 1-2.
Manzeke, G., Mtambanengwe, F., Nezomba, H., and Mapfumo, P. (2014). “Zinc fertilization influence on maize productivity and grain nutritional quality under integrated soil fertility management in Zimbabwe,” Field. Crop. Res. DOI: 10.1016/j.fcr.2014.05.019
Medina, E., Paredes, C., Bustamante, M., Moral, R., and Moreno-Caselles, J. (2012). “Relationships between soil physico-chemical, chemical and biological properties in a soil amended with spent mushroom substrate,” Geoderma 173, 152-161. DOI:10.1016/j.geoderma.2011.12.011
Medina, E., Paredes, C., Perez-Murcia, M., Bustamante, M., and Moral, R. (2009). “Spent mushroom substrates as component of growing media for germination and growth of horticultural plants,” Biores. Technol. 100(18), 4227-4232. DOI:10.1016/j.biortech.2009.03.055
Michalak, I., Tuhy, Ł., Saeid, A., and Chojnacka, K. (2013). “Bioavailability of Zn (II) to plants from new fertilizer components produced by biosorption,” Int. J. Agron. Plant. Prod. 4, 3522-3536.
Michalak, I., Chojnacka, K., Dobrzański, Z., Górecki, H., Zielińska, A., Korczyński, M., and Opaliński, S. (2011). “Effects of macroalgae enriched with microelements on egg quality parameters and mineral content of eggs, eggshell, blood, feathers and droppings,” J. Anim. Physiol. Anim. Nutr. 95(3), 374-387. DOI: 10.1111/j.1439-0396.2010.01065.x
Muller, C., Elliott, J., and Levermann, A. (2014). “Food security: Fertilizing hidden hunger,” Nature Clim. Change. 4(7), 540-541. DOI:10.1038/nclimate2290
Murakami, T., Nishikiori, T., Nohira, T., and Ito, Y. (2003). “Electrolytic synthesis of ammonia in molten salts under atmospheric pressure,” J. Am. Chem. Soc. 125(2), 334-335. DOI:10.1021/ja028891t
Murgia, I., Arosio, P., Tarantino, D., and Soave, C. (2012). “Biofortification for combating ‘hidden hunger’ for iron,” Trends. Plant. Sci. 17(1), 47-55. DOI:10.1016/j.tplants.2011.10.003
Orluchukwu, J., and Adedokun, O. (2014). “Comparative effects of poultry manure and spent mushroom substrate on the growth and yield of pineapple (Ananas comosus) in Nigeria,” Afr. J. Agric. Res.9(26), 2041-2044. DOI:10.5897/AJAR2013.8079
Phan, C., and Sabaratnam, V. (2012). “Potential uses of spent mushroom substrate and its associated lignocellulosic enzymes,” App. Microb. Biotechnol. 96(4), 863-873. DOI:10.1007/s00253-012-4446-9
Polish Ministry of Agriculture and Rural Development. (2008). “Act of fertilizers and fertilization,” Journal of Laws No. 119 Pos. 774, Warsaw, Poland
Putra, W., Kamari, A., Yusoff, S., Ishak, C., Mohamed, A., Hashim, N., and Isa, I. (2014). “Biosorption of Cu (II), Pb (II) and Zn (II) ions from aqueous solutions using selected waste materials: Adsorption and characterisation studies,” J. Encapsulation. Adsorpt. Sci. 4, 25-35. DOI:10.4236/jeas.2014.41004
Qi, B., and Aldrich, C. (2008). “Biosorption of heavy metals from aqueous solutions with tobacco dust,” Biores. Technol. 99(13), 5595-5601. DOI:10.1016/j.biortech.2007.10.042
Ribas, L., De Mendoncca, M., Camelini, C., and Soares, C. (2009). “Use of spent mushroom substrates from Agaricus subrufescens (syn. A. blazei, A. brasiliensis) and Lentinula edodes productions in the enrichment of a soil-based potting media for lettuce (Lactuca sativa) cultivation: Growth promotion and soil bioremediation,” Biores. Technol. 100(20), 4750-4757. DOI:10.1016/j.biortech.2008.10.059
Shi, X., Yuan, X., Wang, Y., Zeng, S., Qiu, Y., Guo, R., and Wang L. (2014). “Modeling of the methane production and pH value during the anaerobic co-digestion of dairy manure and spent mushroom substrate,” Chem. Eng. J. 244, 258-263. DOI:10.1016/j.cej.2014.02.007
Sulaymon, A., Ebrahim, S., and Mohammed-Ridha, M. (2013). “Equilibrium, kinetic, and thermodynamic biosorption of Pb (II), Cr (III), and Cd (II) ions by dead anaerobic biomass from synthetic wastewater,” Environ. Sci. Pollut. Res. 20(1), 175-87. DOI:10.1007/s11356-012-0854-8
Toptas, A., Demierege, S., Mavioglu Ayan, E., and Yanik, J. (2014). “Spent mushroom compost as biosorbent for dye biosorption,” CLEAN-Soil, Air, Water. DOI: 10.1002/clen.201300657
Tuhy, Ł., Samoraj, M., and Chojnacka, K. (2013). “Evaluation of nutrients bioavailability from fertilizers in in vivo tests,” Interdiscip. J. Eng. Sci. 1(1), 10-13.
Wiśniewska-Kadżajan, B., and Jankowski, K. (2013). “The fertilization effect of permanent meadows with spent mushroom substrate on the up taking of manganese, copper and zinc by the meadow sward,” J. Ecol. Eng. 14(2), 68-73. DOI:10.5604/2081139X.1043187
Witek-Krowiak, A. (2012). “Analysis of temperature-dependent biosorption of Cu2+ ions on sunflower hulls: Kinetics, equilibrium and mechanism of the process,” Chem. Eng. J. 192, 13-20. DOI:10.1016/j.cej.2012.03.075
Witkowska, Z., Chojnacka, K., Korczyński, M., Świniarska, M., Saeid, A., Opaliński, S., and Dobrzański, Z. (2014). “Soybean meal enriched with microelements by biosorption – A new biological feed supplement for laying hens. Part I. Performance and egg traits,” Food. Chem. 151, 86-92. DOI:10.1016/j.foodchem.2013.11.023
Wuest, P., Fahy, H., and Fahy, J. (1995). “Use of spent mushroom substrate (SMS) for corn (maize) production and its effect on surface water quality,” Compost. Sci. Util. 3(1), 46-54. DOI:10.1080/1065657X.1995.10701768
Zhan, R., and Duan, Z. (2012). “Study on compound substrate properties with spent mushroom compost and cattle manure compost and effects on the growth of seedlings,” Agric. Sci. Technology-Hunan. 13(1), 149-154.
Zhu, H., Sun, L., Zhang, Y., Zhang, X., and Qiao, J. (2012). “Conversion of spent mushroom substrate to biofertilizer using a stress-tolerant phosphate-solubilizing Pichia farinose FL7,” Biores. Technol. 111, 410-416. DOI:10.1016/j.biortech.2012.02.042
Article submitted: February 13, 2015; Peer review completed: May 2, 2015; Revised version received: July 8, 2015; Accepted: July 9, 2015; Published: July 27, 2015.
DOI: 10.15376/biores.10.3.5709-5719
